
CALCITE
Calcite is an extremely common carbonate, very important as a rock former, occurring in sediments and in igneous, metamorphic and sedimentary rocks. It is an important ore, with hundreds of uses in industry, such as in the manufacture of cement, as mineral filler in cosmetics, paints and rubbers, in steel, metallurgy, agriculture and many others.
Under the microscope, calcite in large, well-formed crystals (spatic) shows cleavage and twins; microcrystalline calcite (micrite) has no cleavage or twins. Macroscopically, it can be any color from black to white, passing through the entire rainbow; it can be zoned or banded. The crystals, even easily formed at room temperature, have more than 2400 combined crystal forms. The largest single crystals known were 7x7x2 and 6x6x3m and weighed 250 tons. Calcite has dozens of different habits. Very useful in its recognition is its strong effervescence with cold diluted (~10%) hydrochloric acid– just a drop is enough to obtain the effect. Calcite is trimorphic with aragonite and vaterite and forms a series with rhodochrosite. It can contain Fe, Mg, Mn, Zn and Co. Calcite has at least 20 varieties.
Calcite can fluoresce in red, blue, yellow, and other colors under both short-wave and long-wave UV light. It is phosphorescent, cathodoluminescent and thermoluminescent, but rarely triboluminescent.
1. Characteristics
Crystal system: Trigonal scalenohedral.
Color: All. From black to white, passing through the rainbow.
Habit: Dozens of different habits.
Cleavage: Rhombohedral, perfect {10-11}, with angles of 60 and 120 degrees to each other.
Tenacity: Brittle
Twinning: Polysynthetic and pressure twins, very common.
Fracture: Conchoidal.
Mohs Hardness: 3
Parting: No.
Streak: White.
Lustre: Vitreous, pearly on cleavage surfaces.
Diaphaneity: Transparent.
Density (g/cm³): 2.7
2. Geology and Deposits
Calcite is the most common carbonate, it can occur in practically all types of sediments and rocks, in the latter either as a primary or secondary mineral. In limestone, marble and carbonatite, it is the main rock-forming mineral.
It is very common as an accessory and can fill veins that cross rocks. It is a very common mineral associated to metallic ore minerals, in this case called “gangue”. The list of possibilities is very long; calcite can always be present because of its ease in dissolving and reprecipitating.
3. Mineral Associations
Calcite occurs in so many different paragenesis, as a primary or secondary mineral, that any list of associated minerals will always be incomplete. Calcite basically occurs in association with any other mineral.
4. Transmitted Light Microscopy
Refraction indices: nε: 1.486 no: 1.658
PLANE POLARIZED LIGHT – PPL
Color / Pleochroism: Colorless, has no color or pleochroism.
Usually it is a bit darker, grayer, than colorless silicates like quartz and feldspars.
Relief: Relief varies from low to moderate to high every 90º when turning the stage in crystals with well defined cleavage. This phenomenon has been called “relief pleochroism” or “chagrin change” and is typical of rhomboedral carbonates (calcite, dolomite, aragonite, siderite, rhodochrosite and magnesite).
When microcrystalline, these carbonates do not show “relief pleochroism” or it is very difficult to perceive.
Cleavage: Rhombohedral {10-11} perfect in three directions. In the crystals there are two cleavages that intersect at angles of 60º and 120º.
When calcite is microcrystalline (“micrite”), no cleavage is observed.
Habits: Normally anhedral in thin section. When it crystallized in cavities, such as vesicles of volcanic rocks, it can be idiomorphic in crystals called “dog teeth” (= scalenohedral). Macroscopically it can show many hundreds of habits and forms.
CROSSED POLARIZED LIGHT – XPL
Birefringence and Interference Colors: Extreme birefringence, 0.172: very high colors, 4th order.
They are cream, brownish, with colored dots and bands.
Extinction: Symmetrical in relation to the two cleavages (the vertical string of the crosshair forms the bisector of the angle between the two cleavages in the extinction position). In the extinction position there is birefringent calcite dust generated by the polishing.
Elongation sign: Does not apply as calcite may be elongated in several directions.
Twins: On {01-12}. They are very common in large crystals and appear as straight, colored, parallel bands. These twins can often be seen even at PPL. The abundance of twins characterizes calcite; dolomite is practically untwinned.
Zoning: No.
CONVERGENT LIGHT
Character: U(-), but can be anomalously biaxial, mainly in metamorphic rocks.
2V angle: No. When anomalous from 0 – 15º.
Alterations: Calcite is very resistant, but it can be easily dissolved, allowing substitutions (pseudomorphoses!) for other minerals in spaces formerly occupied by calcite crystals.
May be confused with: other common carbonates such as dolomite, siderite, magnesite, rhodochrosite and aragonite. Among these, calcite is the carbonate with the most twins. But other analytical techniques are needed to confirm trigonal carbonates. Under a microscope it is only possible to identify these different carbonates with the use of a universal stage, a technique that requires a lot of experience.
Dolomite is more often idiomorphic than calcite.
Titanite shows higher relief, yellowish to brown colors, faint pleochroism and is biaxial.





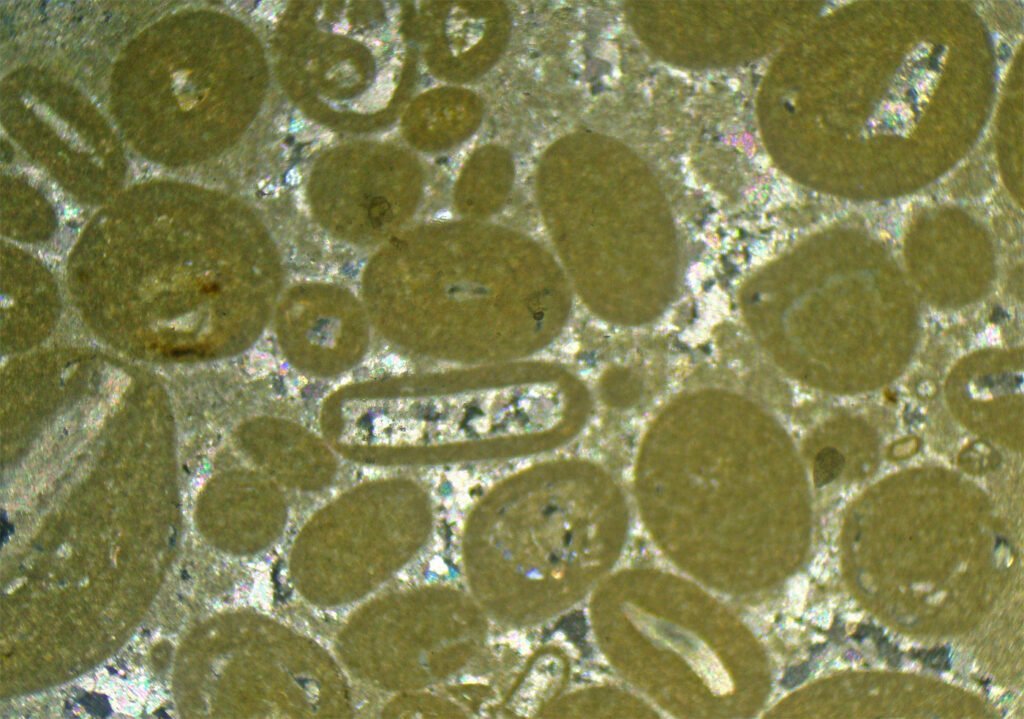





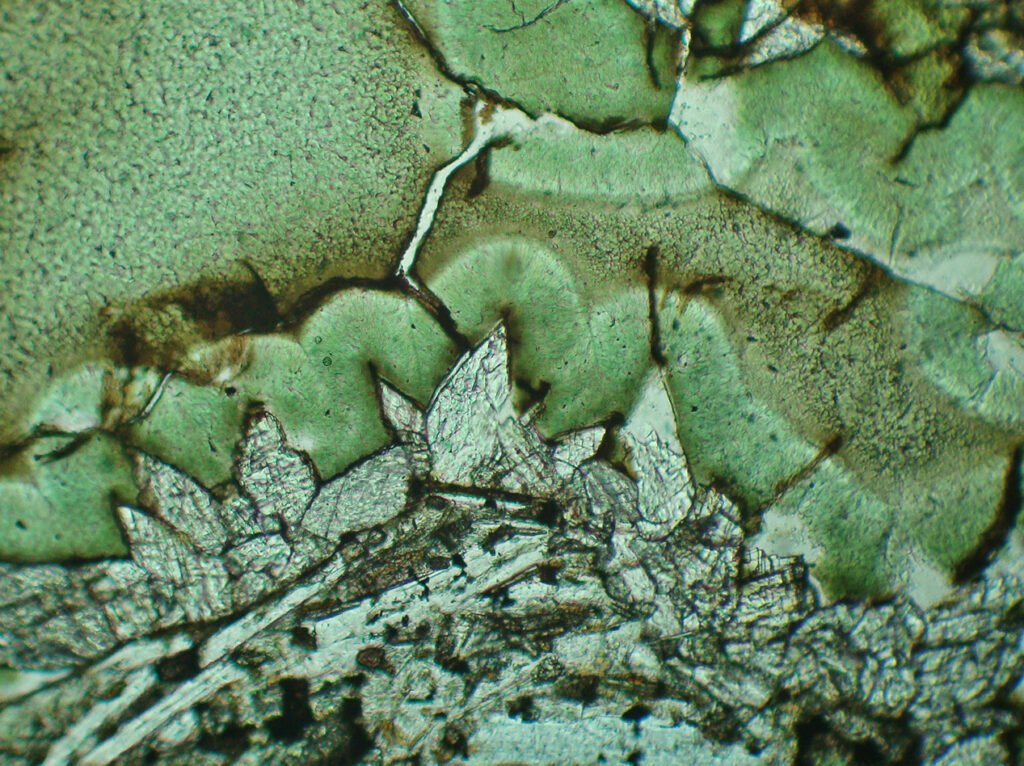

5. Reflected Light Microscopy
Carbonates, including calcite, are easy to recognize under Reflected Light. The situation is the same as for Transmitted Light: the mineral is identified as a rhombohedral carbonate, but it is not known which one it is, as in addition to calcite it could be dolomite, siderite, magnesite, rhodochrosite and others. Calcite usually is characterized by a large number of twins.
Sample preparation: the polishing of calcite is very simple, it is easy and quick to obtain. It is of excellent quality.
PLANE POLARIZED LIGHT – PPL
Reflection color: Dark gray.
Pleochroism: Very strong in shades of gray, easy to observe.
Reflectivity: Very low (~4%)
Bireflectance: No.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: Very strong anisotropy, very evident, in gray. It’s masked by the internal reflections, but it’s usually easy to see.
Internal reflections: Generalized, clear, white to milky.
May be confused with: other carbonates, which have practically the same characteristics under the Reflected Light microscope.
General Characteristics:
Rhombohedral cleavage is easily visible when the grains are large.
Polishing pits are present when crystals are large.
Polysynthetic twins are very conspicuous, easy to see, both in PPL and in XPL. If the carbonate has many twins, it is probably calcite.





