HEMATITE
Hematite – Fe2O3 – is a very common oxide. It is the main iron ore, as it contains approximately 70% iron.
Hematite is one of several iron oxides and forms a complete solution (isomorphism) with ilmenite (FeTiO3) at temperatures above 950°C. At low temperatures there is still a very low degree of mixing with ilmenite and also with MgTiO3. The Ti contents registered in hematites are due to this mixture. FeO contents must originate from mixed magnetites. Hematite is dimorphous with maghemite.
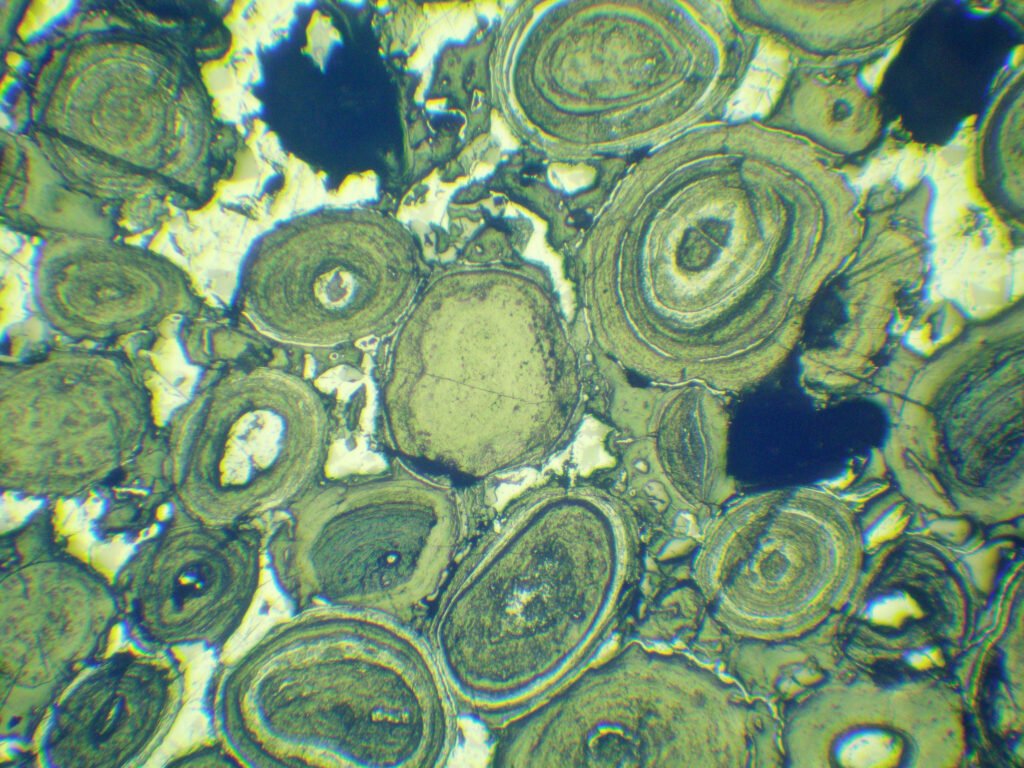
Hematite crystals are rare, reaching up to 13 cm and crystallizing as complex rhombohedral, pseudo-cubic, prismatic and rarely scalenohedral forms, with triangular striations at {0001} and {10-11}. Hematite can show lamellar and interpenetration twins on {0001} and {10-11}. Parting may occur in these orientations due to the twins. Common habits of hematite are fine tabular, micaceous and flattened, usually forming rosettes. Very common are oolitic, massive, radiated fibrous, concretionary, columnar, earthy, reniform, botryoidal or stalagtitic hematite masses.
There are ten varieties of hematite. Among them, “kidney ore” are globular, botryoidal, reniform and mamelonated forms; “martite” are pseudomorphs of hematite over magnetite; “specularite” is a hematite in tabular crystals with an exceptional (specular) luster; “titanohematite” is a variety of titanium-rich hematite; “tiger iron” is a metamorphosed sedimentary deposit, a banded ironstone rock, with alternating layers of gray hematite and red chert, chert or tiger’s eye quartz. The iridescence sometimes exhibited by hematite is formed by “turgite”, a mixture of hematite and goethite, sometimes described as hydrated hematite.
1. Characteristics
Crystal system: Trigonal scalenohedral.
Color: Steel gray to black when massive or in crystals. Red on fine or earthy aggregates. Tarnishes to iridescent.
Habit: See above.
Cleavage: No.
Tenacity: Brittle, elastic in thin flakes.
Twinning: See above.
Fracture: Irregular, subconchoidal.
Mohs Hardness: 5 – 6
Parting: See above.
Streak: Bright red to brown/reddish.
Lustre: Metallic, specular, submetallic to dull.
Diaphaneity: Opaque, transparent in very thin flakes.
Density (g/cm³): 5.26
2. Geology and Deposits
Hematite is found in magmatic, hydrothermal, metamorphic and sedimentary geological environments. Hematite crystallizes in hot springs, lake bottoms, volcanic environments, weathering (soil) and many others. It forms the “banded iron formations” (BIFs) and is generally responsible for the red colors of many geological materials (soils, sands, etc.).
It is an accessory mineral in felsic igneous rocks. Occurs in high-temperature hydrothermal veins, in contact metamorphic rocks, as cement in sedimentary rocks and as a major constituent in oolitic iron formations. The presentation of all deposits is beyond the scope of this approach.
3. Mineral Associations
Hematite is associated with a very large number of minerals, there is no typical paragenesis.
4. Transmitted Light Microscopy
Refraction indices: nω: 3.150 – 3.220 nε: 2.870 – 2.940
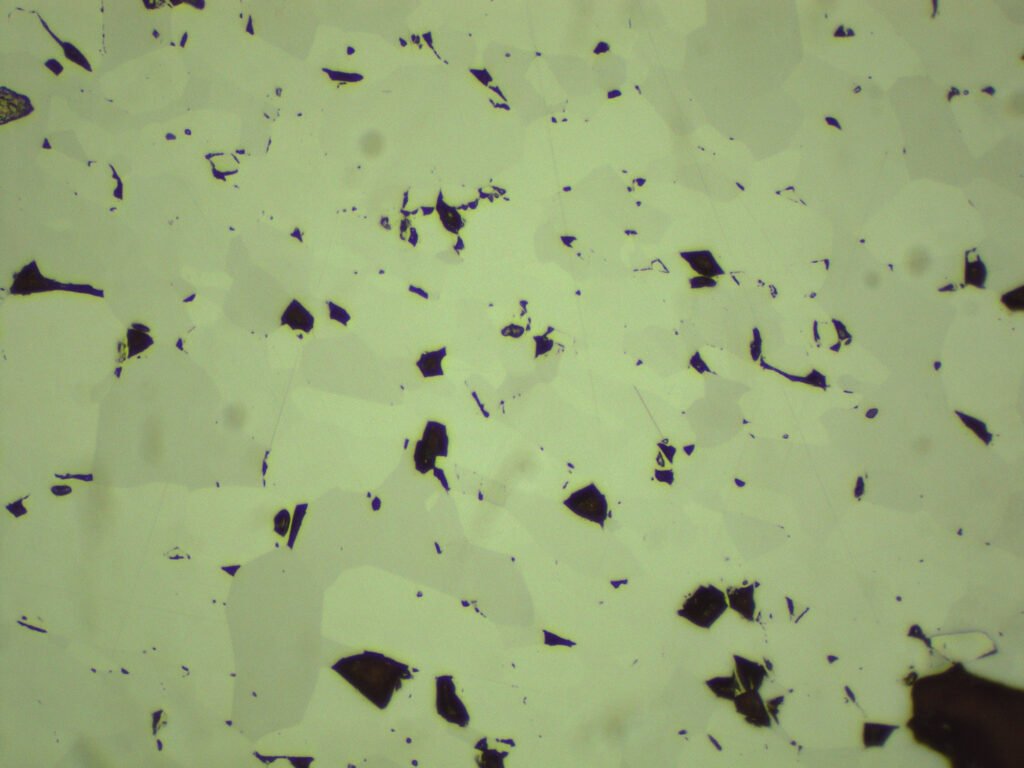
PLANE POLARIZED LIGHT – PPL
Color / Pleochroism: Deep red-brown in very small crystals or along edges, with brownish-red to yellowish-red or brown pleochroism. But generally hematite is opaque, even on sheets less than 30 microns thick.
Relief: Very high, extreme.
Cleavage: No.
Habits: Fine tabular, micaceous and flattened, usually forming rosettes. Very common are oolitic, massive, radiated fibrous, concretionary, columnar, earthy, reniform, botryoidal or stalagtitic hematite masses.
CROSSED POLARIZED LIGHT – XPL
Birefringence and Interference Colors: Maximum birefringence of 0.280: high order colors (some kind of light cream).
Extinction: Paralell.
Elongation sign: ES(-), generally impossible to determine.
Twins: No information about it.
Zoning: No information about it, probably none.
CONVERGENT LIGHT
Character: U(-), generally impossible to determine.
2V angle: No.
Alterations: often hematite is a product of alteration. Can alter to goethite and siderite.
May be confused with: many other opaque minerals, as the red reflections only occur occasionally. It is necessary to use another analytical technique for its identification, like Reflected Light Microscopy.


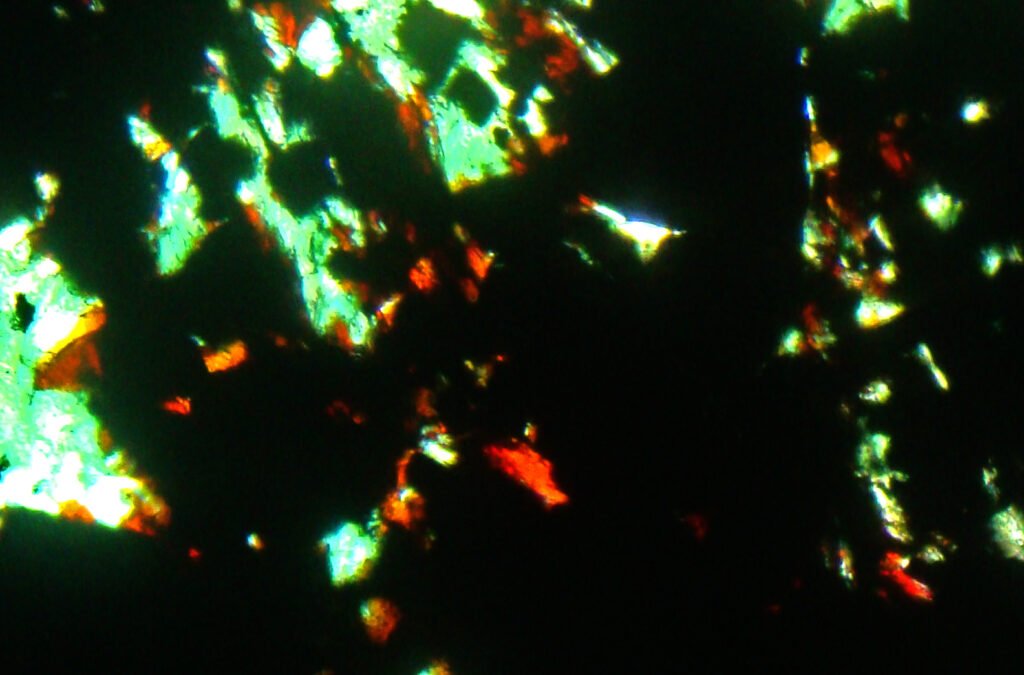
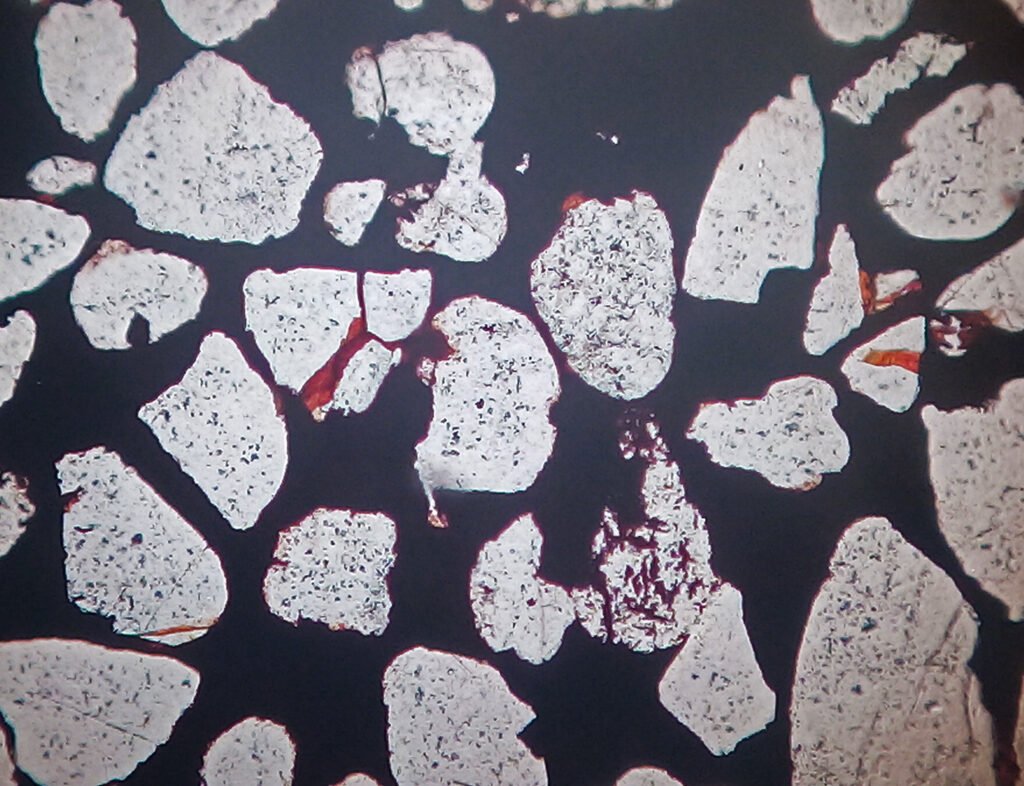
5. Reflected Light Microscopy
Sample preparation: Hematite has a very high hardness and is one of the most difficult ore minerals to polish. Hematite is much harder than magnetite and ilmenite, somewhat less hard than cassiterite. and approximately as hard as pyrite. After a carefully executed thinning, with a lot of patience and twice or triple the time needed for other minerals, an excellent polishing is achieved. Sections parallel to {0001} are more difficult to polish than sections perpendicular to them. Especially difficult is the polishing of the red mamelonated and botryoidal aggregates (“Glaskopf” in German = glass head) and the aggregates formed by long parallel fibers, which keep coming loose during polishing, ruining the polishing of the entire section.
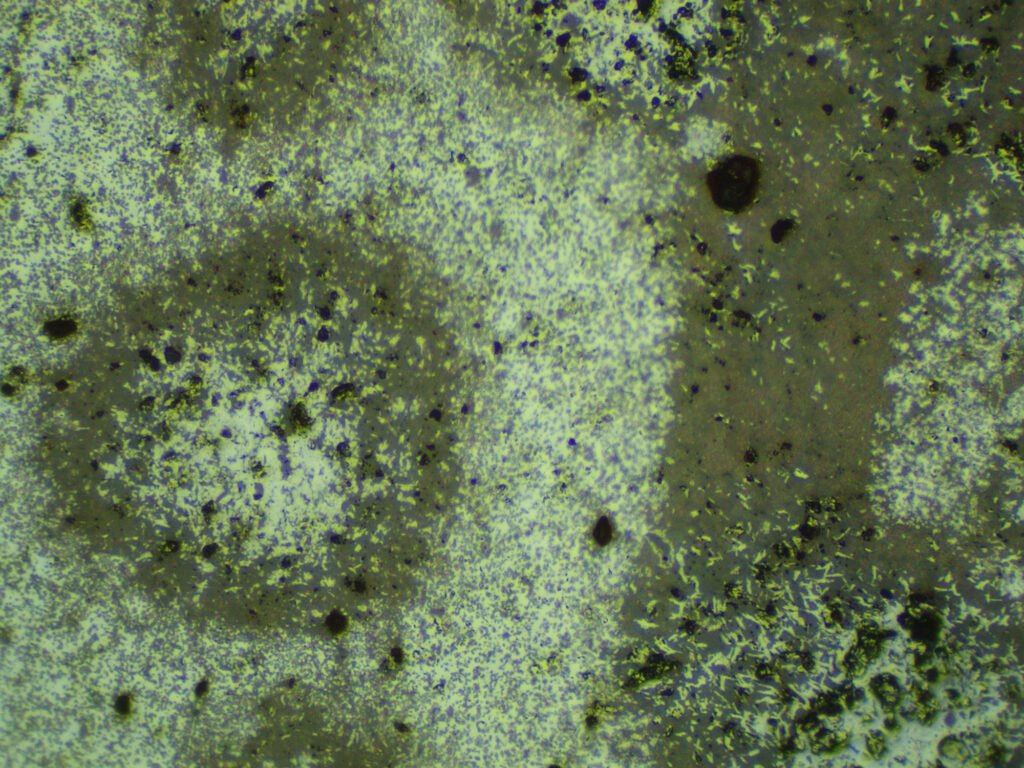
PLANE POLARIZED LIGHT – PPL
Reflection color: White with a blue tint. If titaniferous it is more gray-white.
Compared to the color of pyrite, the color of hematite is light bluish gray.
Compared with the colors of magnetite, ilmenite and cuprite, the color of hematite is white.
Compared to the color of chalcocite, the color of hematite is slightly brownish.
Pleochroism: No.
Reflectivity: 26 – 29.64%. If with Ti, the reflectivity is greater.
Bireflectance: Distinct but faint. If titaniferous the bireflectance is greater.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: Distinct anisotropy in shades of grey-blue to grey-yellow. Well-defined extinction.
Internal reflections: Sparse, rare, small, deep blood red. The amount of reflections depends on the polish: the better the polish, the less reflections there will be.
May be confused with: Usually recognition of hematite is easy. Characteristics are high hardness, poor polishing, weak but sharp anisotropy, bluish-white color and sparse red internal reflections.
Lepidocrocite is similar; in very fine-grained aggregates this differentiation may be impossible.
General Characteristics:
Grain shape: generally hematite is idiomorphic, tabular or fine tabular according to (001), but size, shape and texture depend very much on the conditions of formation of hematite and, therefore, vary a lot. Hematite formed at high temperatures have short shapes and is compact. Hematite from environments with contact metasomatism are often practically isometric. In intrusive ultramafic bodies (diabases) hematite can form very fine, dispersed scales and without clear development of faces. In some cases, hematite appears in mamelonated forms or in long fibers; in this case it is easy to confuse it with lepidocrocite. In deposits that have suffered intense tectonism, thin plates are initially formed, which by recrystallization later give rise to oval bodies, little intergrown with each other. The plates are arranged parallel in the rock, exactly like micas in mica-schists, enveloping and skirting inclusions and idioblasts. The tabular crystals of hematite may be intensively deformed and bent even in a virtually undisturbed rock.
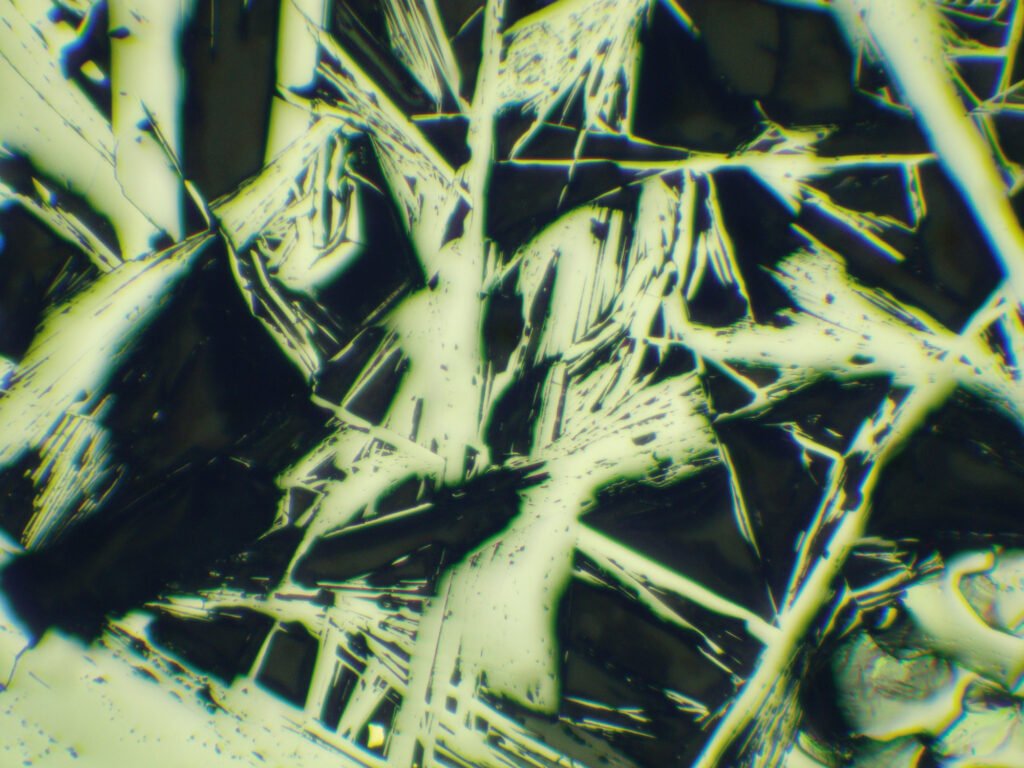
Twinning: lamellar twins according to {10-11} are very frequent and visible, especially in CPL. A smaller amount of the twins originated during growth; most of the lamellae are pressure lamellae, originated by tectonic deformations, always occur in groups and normally according to several faces of {10-11}. The lamellae can occur according to different systems and can show transverse twins. The many macroscopically recognized twins in hematite crystals are unimportant in microscopy.
Partitions {0001} and {10-11} are often visible under the microscope, especially in hematites that have undergone tectonic stresses.
Translations to {0001} are also frequent. Very often they occur side by side with the formation of twin lamella by sliding on {10-11} in the plastic deformation of hematite. This explains the ripples observed in hematites of many occurrences. But in many occurrences of strong tectonic impact these translations are not observed because recrystallization occurred later.
Unmixing: Ilmenite and hematite have complicated mix-up relationships. Up to approximately 10% FeTiO3 remains in solution, giving hematites with a browner trace, rarer internal reflections, and lower reflectivity. In some hematites there are acicular unmixings of rutile, following 3 different orientations. Tiny (>1μm) granular unmixes of cassiterite may also occur. High-gloss and intense hematites may show tiny parallel acicular unmixes(?), observable only very carefully, with oil immersion and not exactly crossed nicols; it can be micro twin lamellae.
“Titanohematite” or “white ilmenite” is hematite with up to 10% FeTiO3 in solid solution. Its reflectivity is lower, bireflectance and anisotropy are stronger. They always have lamellar twins and do not show red internal reflections. If the FeTiO3 content is above 10%, oriented intergrowths of ilmenite and hematite-Ti are formed. Small hematite-Fe lamellae in ilmenite may occur.
Substitutions of magnetite for hematite are a common product of contact metamorphism in intrusive and extrusive rocks, but are found in other situations as well. Very well-developed pseudomorphosis often form. In this case, there may be remnants of magnetite in the hematite, which explains the magnetism of these hematite. The replacement of hematite by pyrite occurs in extrusive-hydrothermal formations with some frequency, especially hematite impregnations in tufaceous breccia, where all stages of transformation can be observed. Normal weathering leads to the formation of limonite, but very slowly. Rare are substitutions of hematite for siderite or chalcopyrite.
Martitization: hematite replaces magnetite, a process called martitization. The process can form large hematite crystals from magnetite. In some cases the process results in large idiomorphic hematite crystals with some scattered remnants of magnetite, which explains the magnetism of these hematites. On the other hand, hematites with polar magnetism of exactly Fe2O3 composition without inclusions of magnetite or maghemite are not rare; its magnetism has been explained by some authors by the impact of lightning.
Inclusions of hematite in chromite may occur.
Inclusions in hematite may be rutile with granular or acicular habit.
Exsolution intergrowths can occur from hematite with ilmenite, magnetite, rutile, cassiterite and franklinite. Oriented intergrowths of some of these minerals with hematite were not necessarily formed by exsolution.