ILMENITE
Ilmenite – FeTiO3 – is a very common oxide that occurs in many different types of rocks. It is the most important Ti ore and the most common accessory ore mineral after magnetite.
It forms 3 series, with ecandrewsite, geikelite and pyrophanite. Usually, ilmenite contains significant levels of Fe2O3 (hematite), in addition to levels of MgTiO3 (geikielite) and MnTiO3 (pyrophanite). It can form pseudo-octahedral crystals and is usually very weakly magnetic, but when pure it is not magnetic.
About varieties: “iserine” are rounded ilmenites found in the placer deposit of the river Iser, in the Czech Republic. “Menaccanite” and “picroilmenite” are magnesium-rich varieties of ilmenite. “Washingtonite” is an iron-rich ilmenite found in Washington (CT, USA).
In altered igneous rocks, ilmenite can be recognized by its white alteration product, leucoxene, which is a submicroscopic association composed of Fe-Ti oxides, rutile, anatase, titanite, perovskite, titaniferous magnetite and especially ilmenite.
1. Characteristics
Crystal system: Trigonal rhombohedral.
Color: Iron black, brownish black, dark reddish brown, brownish gray.
Habit: Massive or granular, skeletal, rarely in thick tabular crystals, up to 25 cm.
Cleavage: No.
Tenacity: Brittle.
Twinning: On {0001}, simple, {10-11} lamellar.
Fracture: Conchoidal, subconchoidal.
Mohs Hardness: 5 – 6
Parting: On {0001} and {10-11}.
Streak: Black to brownish-black.
Lustre: Metallic, submetallic.
Diaphaneity: Opaque, in fine flakes deep brown.
Density (g/cm³): 4.70 – 4.79
2. Geology and Deposits
Ilmenite occurs in large volumes in stratified mafic intrusions, at the pyroxene level. In igneous rocks, ilmenite generally occurs as discrete grains. There may be only magnetite in the rock or only ilmenite, but generally both occur, with one or the other predominating. Many mafic igneous rocks contain grains of magnetite + ilmenite, formed by the oxidation of ulvospinel.
Magnesian ilmenite is indicative of a kimberlitic paragenesis and is part of the MARID (mica-amphibole-rutile-ilmenite-diopside) mineral association of glimmerite xenoliths. Manganiferous ilmenite occurs in granitic rocks and in carbonate intrusions and may contain anomalous niobium. Very common is ilmenite as lamellae in hematite, magnetite and orthopyroxenes,
Another common form of ilmenite occurrence is detrital in placer deposits (black sands).
3. Mineral Associations
There is no specific, typical paragenesis for ilmenite as it occurs in many types of igneous, metamorphic and sedimentary rocks.
4. Transmitted Light Microscopy
When very thin, ilmenite is transparent, dark brown, with n=2.33-2.51, uniaxial.
5. Reflected Light Microscopy
Sample preparation: Ilmenite has a high polishing hardness, higher than that of magnetite and somewhat lower than those of hematite and pyrite. Patience and careful fine grinding are required. But ilmenite often shows a better polish than magnetites that occur in the same rock.
PLANE POLARIZED LIGHT – PPL
Reflection color: Brownish-gray or brownish-gray, sometimes with a slight pinkish or violet tinge. With high unmixed content of Fe2O3 the color is much lighter.
Compared to magnetite, the color of ilmenite is darker and more brownish.
Compared to sphalerite, the color of ilmenite is lighter and browner.
Compared with hematite, the color of ilmenite is much darker.
Compared to chromite, the color is lighter and more red-brown.
Pleochroism: No.
Reflectivity: 18.05 – 19.85% With increasing MgTiO3 the reflectivity seems to become lower.
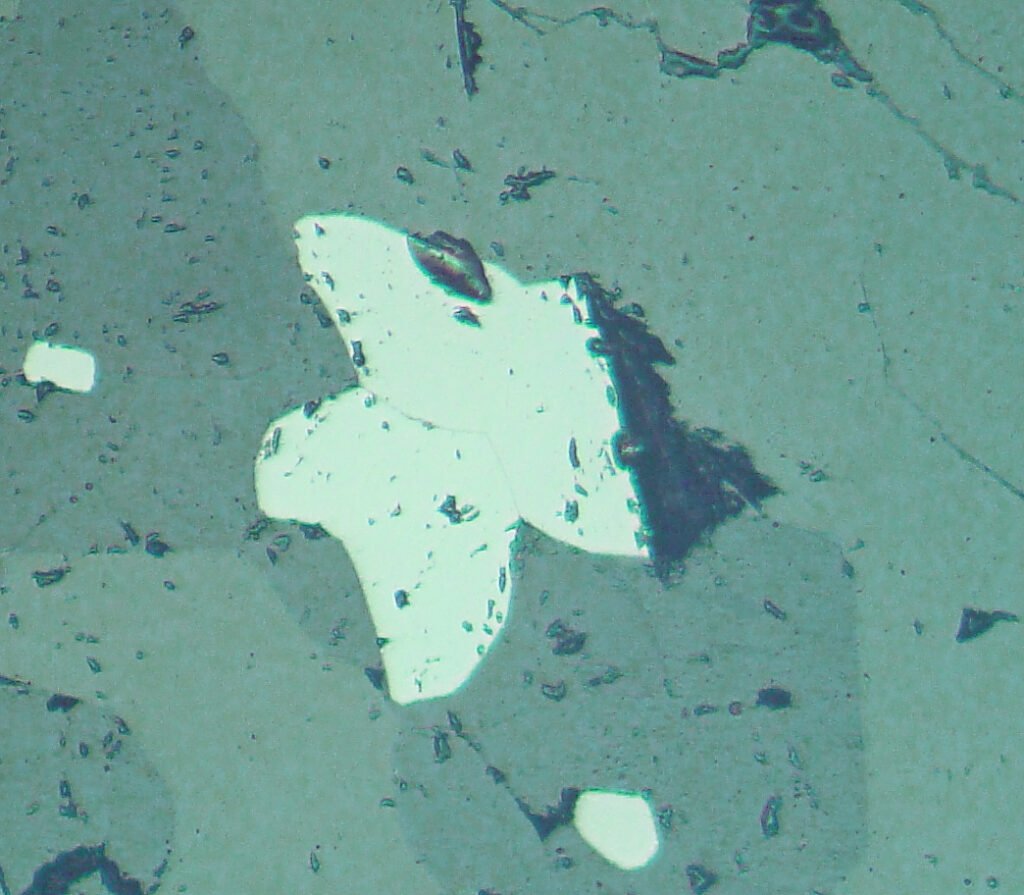
Bireflectance: distinct to very low between lighter brown and pinkish against darker brown. It should be observed in intergranular contacts and next to the twin lamellae.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: Distinct to strong anisotropy in shades of gray, may be colored. Two common colors of anisotropy are light greenish gray and brownish gray. Extinction is parallel in tabular crystals.
Undulating extinction is possible.
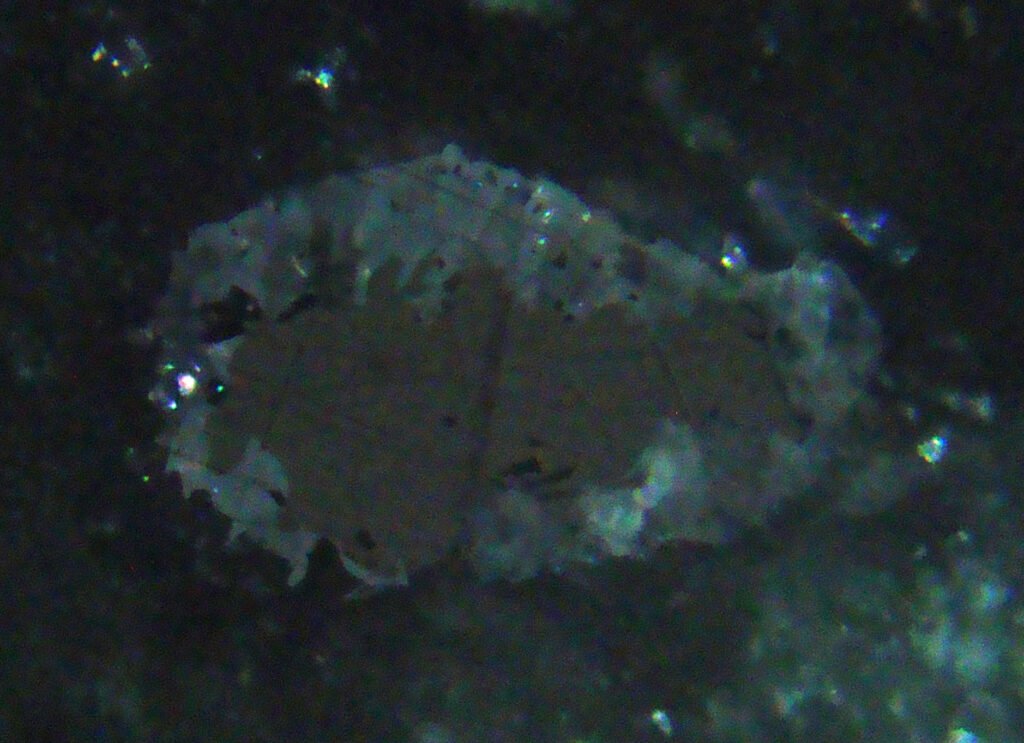
Internal reflections: Very rare, very dark brown. They only appear where the contact with the unmixing lamellae tends to be parallel to the surface of the polished section. In ilmenites with high Mg content, internal reflections are much more common.
May be confused with: several other minerals.
Magnetite and chromite can be very similar in PPL, but are isotropic in XPL. Chromite has lower reflectivity. Basal sections of ilmenite can easily be confused with magnetite, even more so if the magnetite twin lamellae are considered to be unmixings of ilmenite in magnetite.
Geikielite and pyrophanite have more frequent internal reflections.
Rutile may be similar, mostly in small grains and without suitable comparative minerals, but it clearly has internal reflections.
General Characteristics:
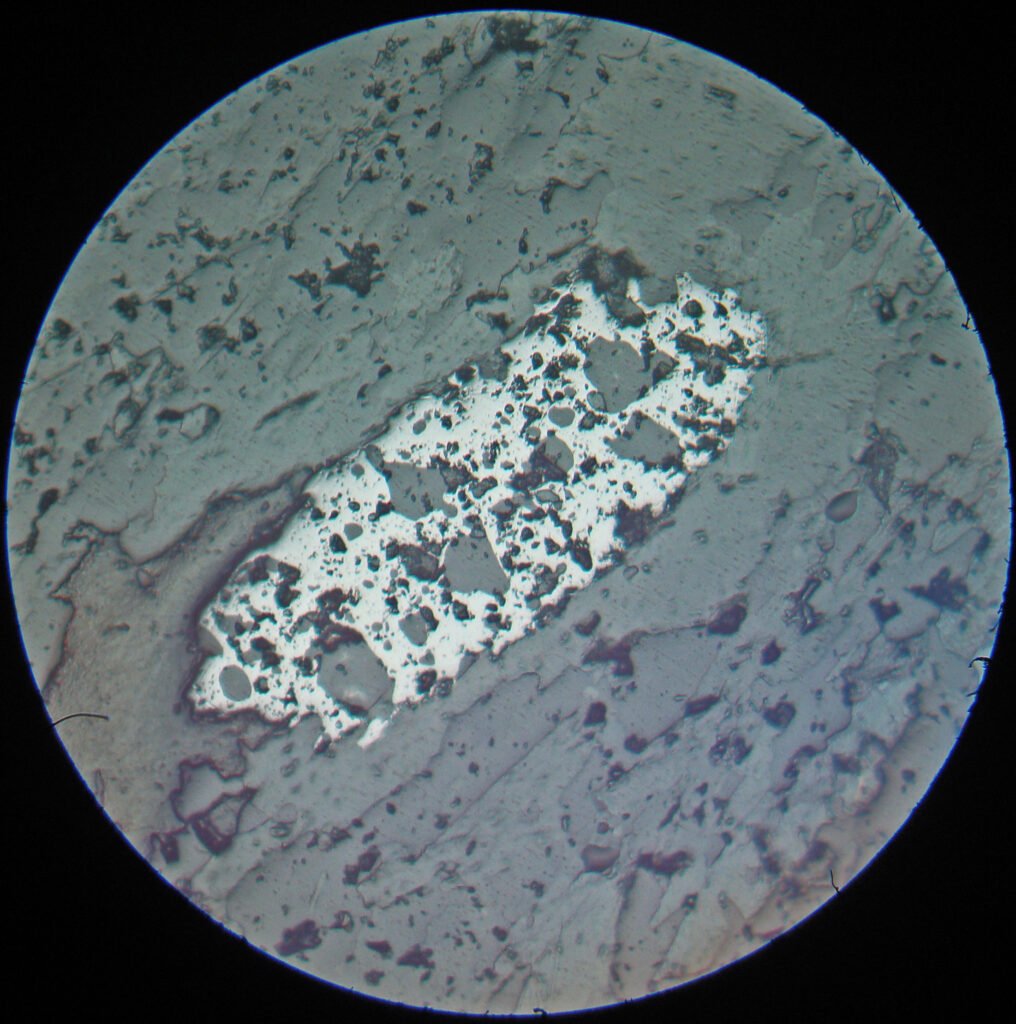
Grain Shape: Ilmenite grains can have many different shapes. Idiomorphy is relatively rare, much rarer than magnetite. Rounded crystals are very common, but can evolve to tabular shapes by (0001), skeletal and myrmekitic. They occur together with magnetites and, in general, it is not possible to distinguish ilmenite from magnetite by shape. Very rarely it is columnar parallel to (0001). In many cases the ilmenites, due to corrosion during the growth of the skeletons, are very irregular or in the form of fragments.
Grain size varies greatly. It is generally roughly equivalent in size to the silicates of the host rock. Porphyroblasts are quite rare, only in Ti-rich amphibolites do porphyroblasts with a typical sieve structure occur. In ilmenite pegmatites and differentiated ilmenite grains reach fist size and above. A regular distribution of grains can be observed in many ores when the ilmenites are with (0001) arranged parallel to the rock texture. Recrystallization textures are not uncommon.
Cleavage does not show, only occasionally may show a partition parallel to (0001), related to unmixings.
Twins, very well-developed, occur in many ilmenites. They can be perceived by bireflectance in PPL and even better by anisotropy in XPL. These are pressure twins in the form of simple lamellae and with the same width parallel to (10-11). Isolated lamellae in one direction or groups of lamellae may occur. The lamellae can follow all sides of (10-11), giving a very fine mesh texture. The thickness of the lamellae is very variable, usually the lamellae are thicker in coarse-grained aggregates. At the intersections of two lamellae, especially in unmixed ilmenites, very often very round (thus rod-shaped) magnetite (± rutile) neoformations occur. The lamellae may be due to growth, but are usually formed by mechanical stresses. The distribution of the twin lamellae shows that they were formed very early, before the unmixings that occurred in the ilmenites. By recrystallization, ilmenite grains, especially those with large volume of twins, can be transformed into grains without any twin lamellae. Evidence of recrystallization is generally quite common.
Regular intergrowths can occur with sulfides such as pyrrhotite, sphalerite, and galena, but are less common features.
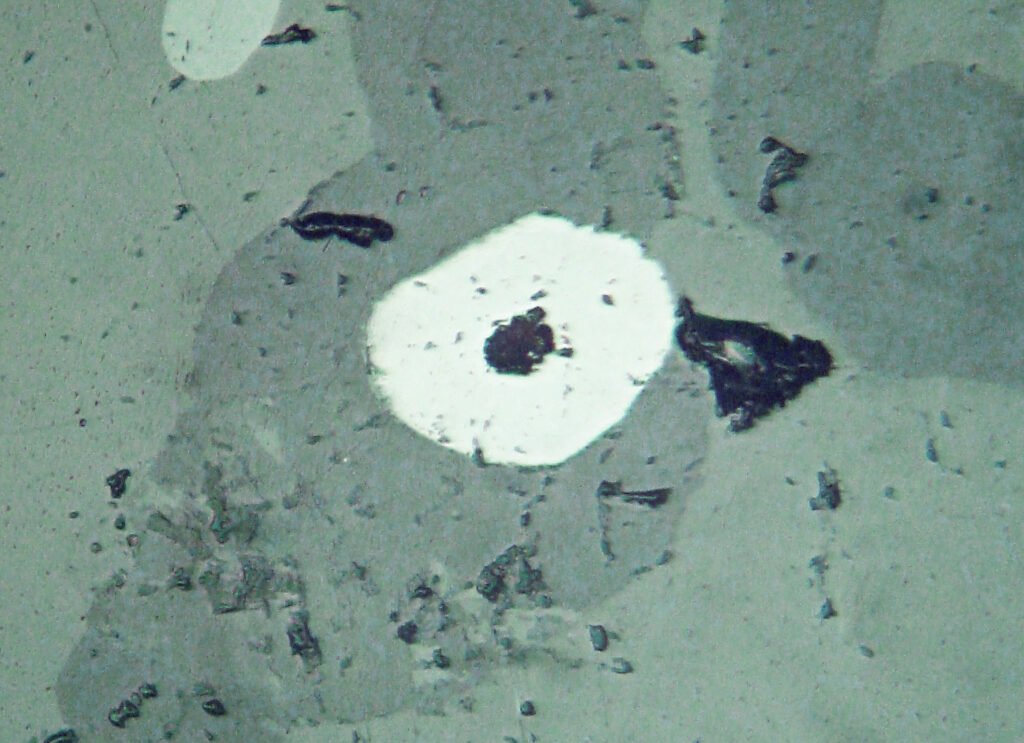
Myrmekites are frequent at the edges of ilmenite grains that are in contact with magnetite in magmatic paragenesis. These myrmekites are well developed and formed by spinel, magnetite and ilmenite.
Zonation is difficult to observe because it is very complicated to carry out a proper chemical attack on ilmenite. In many cases, the zonation becomes perceptible by the distribution pattern of the hematite dismixtures or by other features.
Deformations usually lead to cataclasis. When it is possible to absorb the deformation through small adjustments, the second slip twin lamellae appear (10-11); in rare cases there is also a translation according to (0001).
Reaction rims in the form of myrmekites occur between ilmenite and silicates in gabbros, for example.
Substitutions: ilmenite can be replaced by pyrite, which occurs in many epithermal deposits, especially those of subvolcanic origin. In this process, rutile and other Ti minerals are also formed. Ilmenite is also replaced by rutile, magnetite, hematite, sphalerite, pentlandite and davidite. The latter can develop halos in the ilmenite due to its radioactivity.
Unmixings may be of six types.
Unmixings 1: in many ilmenites there are lenses of exsolved hematite, thicker or thinner, in a very variable amount, arranged parallel to (0001). The size of the lenticular bodies varies greatly and is closely related to the size of the ilmenite grains. The volume of unmixed hematite can be equal to or greater than the volume of ilmenite in the grain; in the latter case there are ilmenite lenses with the same characteristics immersed in hematite. When the volume of unmixing bodies exceeds a certain limit, there are always larger and smaller disks side by side, with no transitional aspects. The coarser discs of hematite in the ilmenite in turn contain smaller discs of ilmenite (or vice versa). In sections parallel to (0001) the disks have very irregular shapes. These features are generated due to the unmixing in stages of the different components of the original melt along the lowering of the temperature (cooling). At least two generations of unmixing lamellae are generated. Mixture crystals low in Fe2O3 often show zonation, related to different crystallization temperatures. FeTiO3-rich mixture crystals are much more frequent than Fe2O3-rich mixture crystals, but all intermediate terms can be observed. The optical characteristics of the grains vary markedly with variation in chemical composition; especially reflectivity increases with increasing hematite content. Unmixed (or remixed) ilmenites with higher Fe2O3 contents are rare, but have been observed in tuffs or lava inclusions, when deep-sea material was rapidly transported upwards and immobilized; these tuff ilmenites serve as guide minerals for certain tuff horizons. Ilmenites that suffered very high temperatures (~1100ºC) generate pseudobrookite, also very frequent in the different occurrences.
Unmixings 2: Some ilmenites from differentiated ores very rich in Al2O3 contain small unmixing bodies, in the form of disks or plates, of corundum. The observation of these unmixings can only be performed with polished sections of superior quality due to the hardness (9) of corundum.
Unmixings 3: in many ilmenites very rich in unmixed hematite, there is a large amount of rutile needles (actually thin plates), arranged obliquely to (0001) in up to 6 distinct groups.
Unmixings 4: some ilmenites show parallelly arranged magnetite inclusions (0001). Inclusions have thin, very uniform, often relatively large, tabular shapes. Its amount is usually well below 10%, it can go up to 15%. Very rarely it is very coarse. These inclusions derive from the unmixing of an original magnetoilmenite (ilmenite + Fe2O3 +FeO). Immediately adjacent to the magnetite lamellae, the ilmenites are markedly darker than in the unblended portions. These unmixed magnetoilmenites are quite frequent; additionally, demixing bodies of magnetite and hematite rarely occur in the same ilmenite. The occurrence of these magnetite inclusions and magnetite rods at the intersections of twin lamellae (see under “twins”) alter the magnetic behavior of ilmenite and are responsible for problems in the use of the ore. In some cases, observed in some granites and in pyrrhotite/pentlandite deposits, ilmenites with 30-50% or more magnetite occur, probably generated by the reduction of the hematite content of ilmenites especially rich in Fe2O3. The thick discoid hematites are transformed into parallel tabular magnetites.
Unmixings 5: Unmixings of tantalite may occur.
Unmixings 6: very rarely occur fusiform unmixings of ilmenite arranged parallel to (0001) inside the ilmenite grains. The properties of these unmixings differ very little from those of the host ilmenite and are analogous to the inclusions of hematite in hematite.
Alteration: Ilmenite is quite resistant to normal weathering processes, so much so that the ilmenites contained in ocean beach sands are often unaltered. However, ilmenite can change in several ways:
(a) Magnetite is formed, usually alongside rutile: there are fusiform bodies, similar to unmixings, parallel to the rhombohedron faces of the ilmenite.
(b) Fe2O3-rich ilmenites decompose into an irregular spotted aggregate of rutile + magnetite. These two processes already start at high temperatures.
(c) Ilmenite forms rutile and hematite by the absorption of oxygen. Depending on the temperature, the products are coarser and more clearly interwoven with the original mineral in an oriented manner. In some Alpine tectonic rifts anatase was formed instead of rutile.
(d) In many rocks the ilmenites have altered at the edges or completely to leucoxene (see introductory text). Normally, anatase is formed, whose crystals are generally not oriented in relation to the ilmenite. More rarely, leucoxene is composed of rutile or brookite. In mixing crystals with demixing lamellae the components can behave in different ways: sometimes the hematite lamellae or the magnetite lamellae were selectively removed and transformed into other minerals.
Alteration of ilmenite in heavy sands of ocean beaches: in many cases the black sands of oceanic placers, once without magnetites, contain in the concentrate more TiO2 than corresponding to the ilmenite formula, without the presence of rutile or other titanium mineral. These are diffuse zones of altered color, parallel to (0001), which are formed by an amorphous compound, practically isotropic of Fe and Ti, very rich in Ti. These zones transition to a finely dispersed rutile leucoxene, sometimes to leucoxene from brookite or anatase. This type of alteration is apparently restricted to tropical and subtropical climates.