MAGNETITE
Magnetite – Fe3O4 – is a very common oxide, occurring in many types of rocks. It has enormous importance as ore of Fe, V and Ti, among others. It participates in the composition of many other types of ores.
Magnetite is the most magnetic mineral known and may contain Ti, Mn, Mg, Zn, Ni, Al, Cr and V. It is part of the Spinel Group and has complicated relationships with ilmenite, hematite, hercynite and ulvöspinel. In plutonic rocks, Ti contents are almost always unmixed as ilmenite, more rarely as pyrophanite or geikielite. It forms a series with jacobsite and another series with magnesioferrite.
Typically presents octahedral crystals, sometimes dodecahedral, very rarely tiny cubes (in oolitic magnetite). A variety of strongly magnetic magnetite is known as “lodestone” (“leading stone”); a somewhat altered magnetite that attracts iron. “Thermite” is a mixture of pulverized magnetite and aluminum which, when burned, produces intense heat, aluminum oxide and molten iron.
Magnetite can alter to martite (= hematite). The transformation is progressive, ranging from magnetites with a minimum of hematite to magnetites entirely transformed into hematite. Martite is no longer magnetic (but the magnetism of the samples varies greatly), the hardness goes up to 6-7, the density remains the same, the luster is just sub-metallic and the trace is now reddish-brown. Pseudomorphoses of martite on magnetite are very frequent. The martitization usually brings problems in the ore dressing due to the decrease in magnetism.
1. Characteristics
Crystal system: Cubic hexaoctahedral.
Color: Black, with shades of brown, gray or steel blue. There may be a yellow-brown film of rust.
Habit: Octahedrons, combined cubic shapes. Granular, massive, skeletal. Crystals up to 25 cm.
Cleavage: No. Striations parallel to [011] on {011}.
Tenacity: Brittle.
Twinning: On {111}, contact twins.
Fracture: Irregular.
Mohs Hardness: 5.5 – 6.5
Parting: {111} excellent, was considered cleavage during centuries.
Streak: Black.
Lustre: Metallic to submetallic, can be dull.
Diaphaneity: Opaque, translucent in fine edges.
Density (g/cm³): 5.17 – 5.18
2. Geology and Deposits
Magnetite is the most important and most common opaque mineral in almost all igneous and metamorphic rocks, generally occurring in small grains. In igneous rocks, it usually occurs as titanomagnetite, with the replacement of some Fe by Ti (Fe2TiO4). Through processes of magmatic segregation or contact metamorphism, it can form economic deposits.
It is common in sediments and sedimentary rocks (“banded iron formations”) and makes up part of the “black sands” or “heavy sands”. It occurs in peridotites, carbonatites and meteorites. It occurs biogenic in organisms, from bacteria to orcas, allowing the orientation of animals.
3. Mineral Associations
Magnetite occurs in igneous rocks, with chromite, ilmenite, ulvospinel, rutile, apatite and silicates. In hydrothermal and metamorphic occurrences, with pyrrhotite, pyrite, chalcopyrite, pentlandite, sphalerite, hematite and silicates. With hematite and quartz in sedimentary occurrences.
There is no diagnostic paragenesis. It is always useful to test an unknown rock with a magnet.
4. Transmitted Light Microscopy
In a thin section under Transmitted Light, magnetite is opaque, there is no passage of light even in extremely thin portions. It is the same behavior as other common opaque minerals such as ilmenite, hematite, pyrite, pyrrhotite and chalcopyrite. As a result, it is not possible to identify magnetite under Transmitted Light, it is necessary to work with polished sections or polished thin sections with a Reflected Light microscope.
The most you can do is use the Oblique Light technique and recognize the color of the mineral. If it is gray, it’s certainly not one of the common sulfides like pyrite, pyrrhotite, and chalcopyrite; it will likely be magnetite, hematite or ilmenite.
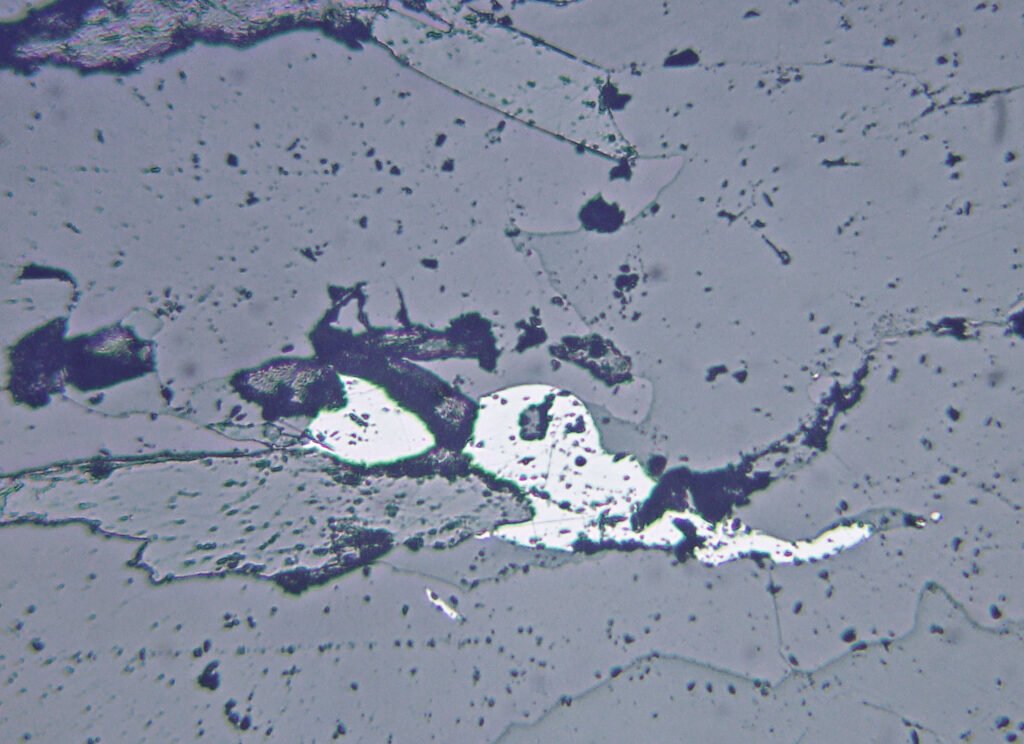
Magnetite is generally xenomorphic (anhedral). In the images below, a less common case, of idiomorphic magnetites in the form of octahedrons and other cubic forms, combined or not. In this rock there are at least two generations of magnetite, one formed by these large crystals and a second one formed by aggregates of smaller crystals, probably of crystals tending to idiomorphic.








5. Reflected Light Microscopy
Sample preparation: Magnetite has high hardness and is difficult to polish. Generally, magnetite has a poor polish, while the associated minerals (silicates, sulphides, etc.) are already polished to a good standard. Insisting on a perfect polish of the magnetite generally produces a high and uncomfortable relief of the magnetite in relation to the less hard associated minerals.
The hardness of magnetite varies with chemical composition and is much lower than the hardness of hematite and pyrite. Pyrrhotite, which often occurs in association, has a much lower hardness. The quality of the polish is still diagnostic: pyrrhotite has a very good polish, while magnetite, of a similar color and habit, shows a low-quality polish. Ilmenite generally shows better polish than magnetite. With some care, pure magnetite acquires an excellent polish.
If martitization is very advanced, polishing becomes very difficult. Some unmixed titano-magnetites are difficult to polish due to the peeling that occurs parallel to (111).
PLANE POLARIZED LIGHT – PPL
Reflection color: Slightly pinkish gray or slightly brownish. The color may change from case to case, without a definite relationship to variations in chemical composition. Variations in the contents of the various minor elements produce colors that overlap randomly. If the magnetite occurs without other opaques for comparison, the color may appear white.
Ti at high and unmixed levels generates a brown-pink color.
Mn makes the color less intense, grayer; very high levels of Mn produce a yellowish-green color. Al and Mg produce yellowish colors. Cr produces colors in a darker gray.
Compared with the color of hematite, magnetite is much darker, tends to brown.
Compared with the color of ilmenite, the color of magnetite is less pinkish.
Compared with the color of sphalerite, the color of magnetite is much lighter.
Pleochroism: No.
Reflectivity: 20.60%
Bireflectance: No.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: Isotropic, but sometimes distinctly anisotropic due to zonation or twins, tectonic stresses or submicroscopic inclusions of another mineral. Stresses generated by blasting and ore treatment can also generate anomalous anisotropy.
Internal reflections: No. Only magnetites with high Mn contents can show some (probably reddish/brown) internal reflections.
May be confused with: several other minerals.
In complex ores, magnetite is easily overlooked, it is important to test the polished section with a magnet or a compass. Especially disturbing is the fact that the reflection color changes a lot.
Ilmenite, which usually occurs in association, is distinctly anisotropic and generally better polished.
Pyrrhotite, which is also frequently associated, has a more yellowish color and strong anisotropy.
Chromite has lower reflectivity, may have internal reflections, but can be very similar.
Sphalerite, if iron-poor, is very similar, just a little darker.
Braunite is distinctly anisotropic.
Jacobsite has deep red internal reflections.
General Characteristics:
Grain shape: magnetite grains can show great tendency to idiomorphy, forming octahedrons, rhombododecahedrons, trapezoidal dodecahedrons and many others. In this case, the sections tend to be triangular, square, trapezoidal and hexagonal. Cubes are quite rare and usually very small. It usually occurs as irregular rounded grains. In volcanic rocks, it can occur in two generations: idiomorphic large grains and small rounded grains, disseminated in the matrix. In magmatic rocks, it occurs as a deuteric mineral from the alteration of biotite and “hornblende”, forming black (opaque) borders around the grains of these minerals. Tabular magnetite crystals can be pseudomorphic on hematite. In massive ores the development is allotriomorphic (anhedral); the grains have little toothed contacts. In deposits that have suffered recrystallization due to regional metamorphism, the contacts can be toothed, but these are rare cases. Rarely, magnetite shows a coloform, reniform or botryoidal habit. The size of the crystals varies from very fine dust to 25 cm, due to the many ways in which magnetite is formed. Ilmenite may exhibit similar habits; under Transmitted Light and Oblique Light it is not possible to distinguish magnetite and ilmenite by habit.
Partition, considered cleavage for many decades, is generally unobservable and occurs only in a few deposits. Lamellar twins can simulate cleavage both macroscopically and microscopically. Spinel unmixing bodies arranged parallel to (100) can generate a pseudo-cleavage.
Deformations are quite common. Magnetite is an early formation mineral and therefore had many opportunities to undergo deformation. Deformations usually generate cataclasis; secundary pressure twin lamellae often form (111). After intense deformation, magnetites easily undergo recrystallization, which is expressed in the textures of magnetites or by intergranular contacts that are not welded (“friable ores”).
Zonation is well developed in almost all magnetites formed by contact metasomatism; occasionally occurs in magnetites of magmatic and hydrothermal origin. Zonation is certainly due, in part, to variations in chemical composition, as small color changes are noticeable even without chemical attack. Other zonations are due to variations in growth velocity. Individual zones can be extremely delicate, very thin. It is often observed that the zoned crystal changed its habit during growth, especially from rhombododecahedron to octahedron. Progressive martitization or other types of substitution often develop preferentially along some isolated areas. In volcanic rock magnetites, there is sometimes a spinel core surrounded by a thick layer of zoned magnetite. In somewhat basic but already metamorphic plutonic rocks, the same occurs with chromite.
Twins parallel to (111) are very common, often lamellar. When the twins are growing, few lamellae are generated. Stresses normally generate numerous and very thin lamellae clusters along all faces of the octahedron in a triangular pattern. In some instances, there are no lamellae, while in others all grains have many lamellae. Macroscopically and microscopically, the lamellae can simulate cleavage!
Oriented intergrowths with ilmenite and hematite may be present not only by unmixing and substitution processes, but also by the individual formation of the two minerals side by side.
Myrmekites are relatively rare, but myrmekites of magnetite and chalcopyrite can occur, as well as myrmekites of magnetite and olivine, sharing the pseudohexagonal axis, which should be interpreted as reaction crowns. Magnetite myrmekites with silicates other than olivine are common especially in gabbros.
Inclusions OF magnetite occur in sulfides.
Inclusions IN magnetite can be hematite, ilmenite, sulfides, awaruite and Spinel Group minerals.
Porphyroblasts of magnetite form easily and can reach significant sizes. Such crystals, always octahedral, are also observed in many ore deposits that have undergone recrystallization.
Alterations are not very common and occur in four main types:
Alteration 1: Magnetite is very resistant to weathering. However, magnetic magnetites (those that attract iron) exposed to the weather for a long time transform into maghemite, in the form of irregular spots, but with a certain preference for (111).
Alteration 2: An occasional change from magnetite is to limonite (goethite and other Fe (hydro)oxides), a process that can occur in magnetite grains on the surface of lava flows.
Alteration 3: Titanomagnetite can change to leucoxene, which shows intense white luminosity under CPL. During the alteration of the unmixed titanomagnetites, the magnetite can be selectively removed/dissolved, leaving a skeleton of lamellae of ilmenite, which is sometimes completely transformed into rutile or anatase (± hematite) and this later to titanite, etc. These features compose relict structures with great petrographic/petrogenetic potential for the study of deposits.
Alteration 4: Magnetite can alter to pyrite in certain environments, such as hot sulfur springs, during propylitization and by mineralizing solutions. The augites, hornblendes and chlorites that occur there are also transformed into pyrite. All stages of the process can be observed, sometimes side by side. The ilmenite skeletons can be preserved, whether or not they have been transformed into rutile or titanite.
Martitization is the process of altering magnetite to hematite. It is very common and occurs in two ways.
Cooling maritization: In the laboratory, the transformation of magnetite into hematite takes place only at high temperatures, but in most deposits it occurred at much lower temperatures. In many cases, martitization must have been the final phase of the processes that generated the deposit; isolated hematite is also formed next to the martitized magnetites. In some deposits in extremely arid regions, martitization is certainly the result of incipient alteration; in these situations the recognition of hematites can be difficult due to their intergrowth with lepidocrocite.
Martitization generally consists of the development of thin plates of hematite, arranged along the octahedral structural planes of the magnetite, starting from the edge of the magnetite, from fractures, holes and twin lamellae, invading the magnetite and replacing it until only isolated remains of the original magnetite remain. Generally, the amounts of hematite arranged in the octahedral planes of the magnetite are approximately the same (and the same size) in all directions. Often, however, especially if the martitization has already started at high temperatures, one of the octahedral directions is preferred, which can generate a pseudomorphosis where the magnetite crystal transforms into a single hematite crystal. In other cases, martitization progresses completely irregularly, even in the form of a network.
The transformation of magnetite into lepidocrocite is very similar and can occur in the same grain. If lepidocrocite occurs alone (unaccompanied by hematite) and in thin lamellae, it will always be confused with hematite. Martitization in magnetites from sulphide deposits, on the other hand, is rare.
Heating martitization: when magnetite is heated under oxidizing conditions, martite also develops. This occurs on the surface of lava flows, in blocks ejected during volcanic episodes, often in ash and lapilli and other similar situations. This martitization has a somewhat different appearance than the cooling martitization, with the lamellae being wider and crossing the entire magnetite crystal or arranging themselves in the magnetite crystal evenly. The typical texture of heating martitization can transform these martites into index minerals of the heavy sand fraction.
Unmixings are very common and occur in nine main types:
Unmixing 1: The original Ti content of magnetite, often high, is usually unmixed as ilmenite (FeTiO3), sometimes in two generations. This unmixing is common in magmatic magnetites, but also occurs in those of pegmatitic-pneumatolitic origin and contact metasomatism. Ilmenite unmixing bodies generally constitute secondary plates (0001), arranged parallel to (111) of the magnetite. Their size varies within wide limits, from 1 mm thick and several cm2 in area to so thin that they are at the limit of detection under the microscope. In addition to these plates or lamellae, rounded grains are formed, also drops, sometimes fine “graphic” aggregates or a very fine network arranged parallel to (100). The form of the unmixing bodies depends on the magnetite grain size, cooling rate, composition and presence of mineralizers. Homogenization of these magnetites with unmixed ilmenite can be seen at all stages in some volcanic rocks. Generally, the unmixing bodies cover the magnetite uniformly, but exceptions are not rare, in which magnetite grains may have an outer crust completely free of ilmenite.
Unmixing 2: Another possible unmix is of pyrophanite (MnTiO3), forming lamellae similar to ilmenite, as well as geikielite (MgTiO3). Pyrophanite is similar to ilmenite, but the internal reflections are blood red and much more frequent. Geikielite is darker than ilmenite. Intermediate compositions between FeTiO3 – MnTiO3 – MgTiO3 are not rare.
Unmixing 3: Spinel (MgAl2O4), is a more frequent unmixing body in magnetites formed at higher temperatures. Perhaps the most frequent unmixing is that of ulvöspinel (Fe2TiO4), which forms very fine networks parallel to (100), called “cloth texture”, very similar to those of ilmenite. As it has the same color as ilmenite, recognizing this mineral is very difficult, generally it is only possible through X-Ray Diffractometry. Unmixing networks formed by ulvöspinel can pass later to ilmenite. Generally, the unmixing of ulvöspinel occurs after the unmixing of the ilmenite in the magnetite. Many ulvöspinel magnetites also contain graphite in the form of spherical bodies or in fine-grained aggregates. In some cases, there are ilmenites and olivines with unmixing bodies of magnetite, which in turn later decomposes into magnetite and ulvöspinel. The ulvöspinel transforms very quickly to ilmenite even with incipient oxidation; in the initial stages the shape of the original unmixing lamellae is still clearly visible, but they are already formed by a zigzag of ilmenite lamellae; a magnetite + ilmenite myrmekite often forms quickly. High levels of ulvöspinel bring problems in processing; the magnetic concentrate can continue with the original Fe and Ti grades of the ore.
Unmixing 4: The most common unmixing bodies of aluminous spinels are hercynite (FeAl2O4) and pleonaste (Fe-rich spinel). They form very fine disks or alignments of small grains arranged parallel to (100) of the magnetite; can also form an edge on the ilmenite unmixing plates. The zoned distribution of spinel in magnetite is much more conspicuous than the zonation of ilmenite unmixing bodies. When there are neighboring grains of ilmenite and spinel, the ilmenite is usually older.
Unmixing 5: In some cases, an unmixing of a very rich Mg spinel occurs, in a texture similar to the unmixings of ulvöspinel.
Unmixing 6: A very rare unmixing is corundum (Al2O3), usually occurring alongside spinel unmixings.
Unmixing 7: Under extremely high temperatures (> 1,000oC) some unmixing of hematite into magnetite occurs. In the laboratory, these unmixings are quite conspicuous; in rocks these unmixing bodies are very rarely observable.
Unmixing 8: Unmixed titanomagnetites are relatively frequent in volcanic rocks and in materials of great depths transported rapidly to the surface (“blue ground” – non-oxidized kimberlites). The spinel content also remains unmixed. These high Ti magnetites have ilmenite-like reflection colors, especially in oil immersion. Magnetites with high Mg and Al contents have a lower reflectivity than that of ilmenite and pure magnetite. The contents of V (often of economic importance) and of FeO without TiO2, which can be present at 5-6% above the formula value, do not appear as an unmixing.
Unmixing 9: magnetite occurs as a unmixture, forming beautiful star-shaped aggregates in many olivines of intermediate composition between fayalite and forsterite as well as in some fayalites. There, [111] of magnetite coincides with the pseudohexagonal axis of olivine.
Substitutions come in seven different types:
Substitutions 1: Due to variations in oxygen pressure or under the influence of reducing solutions, even without raising the temperature, hematite is replaced by magnetite, forming well-developed pseudomorphs. In Russian literature, these pseudomorphs are called “Muschketoffite”. By contact metamorphism or by regional metamorphism at greater depths, this transformation is frequent; the base of the hematite is transformed into one of the faces of the magnetite octahedron, sometimes the process is selective, forming a magnetite crystal with partition on only one of the faces of the octahedron, corresponding to (0001) of the original hematite.
Substitutions 2: the transformation of olivine into serpentine generates a network of magnetite, which forms small aligned grains. The “opaques” of serpentinites, therefore, are usually magnetite. Under these conditions, native iron and awaruite (Ni3Fe) can also be formed.
Substitutions 3: Magnetite can also form from limonite (goethite) and hydrated silicates under low pressure contact metamorphism, therefore mainly in contact with volcanic rocks, but also from carbonates and sulfides.
Substitutions 4: Under incipient alteration, pyrrhotite can transform into magnetite + pyrite, giving rise to fine structures that resemble myrmekitic intergrowths.
Substitutions 5: Oolitic magnetite is formed by replacing hematite and goethite with magnetite. The magnetite replaced the oolites and all the remains of organisms that accompany them in every detail, the presence of metamorphic processes can be excluded.
Substitutions 6: Other minerals that can be substituted for magnetite are pyrrhotite, chromite, pentlandite and heazlewoodite.
Substitutions 7: magnetite can also be substituted by chalcopyrite, pentlandite, cubanite, sphalerite, bornite, lepidocrocite, pyrolusite, pyrrhotite, gold, stannite and galena.