TITANITE
Titanite – CaTiSiO5 – is a very common nesosilicate, an accessory mineral to many types of igneous rocks. A traditional name for titanite, still widely used, is “sphene”. It is a TiO2 ore, an important white pigment. Furthermore, it can be used as a gemstone and for dating by the U-Pb method.
It may contain Al, Fe and F. Furthermore, it contains Nb, Ta, Fe, Mg, Mn, Na, Y, Ce, Sr, V, Zr and Sn as impurities. Forms a series with malayaite, an extremely rare monoclinic nesosilicate that is the Sn analogue of titanite. Titanite has 9 varieties (with Cr, with Ce, with Mn, with Y, with Sn, a triclinic one, etc.). Depending on its content of radioactive elements, it can be metamict.
1. Characteristics
Crystal system: Monoclinic prismatic.
Color: Colorless, brown, black, green, yellow, pink-red.
Habit: Granular, tabular, wedge-shaped, prismatic, compact, massive.
Cleavage: {110} good.
Tenacity: Brittle.
Twinning: On {100}, penetration twins or contact twins, may be lamellar.
Fracture: Subconchoidal.
Mohs Hardness: 5 – 5.5
Parting: Due to twinning, on {221}.
Streak: Reddish white.
Lustre: Adamantine, resinous.
Diaphaneity: Transparent.
Density (g/cm³): 3.48 – 3.60
2. Geology and Deposits
Titanite is very common as an accessory mineral in many types of igneous rocks. In plutonic igneous rocks it is more common in diorites, syenites, monzonites, tonalites and granodiorites, usually associated with a greenish amphibole. Large crystals are found in pegmatites. It also occurs in Alpine Type tectonic fissures.
In volcanic rocks it is more common in phonolites; it does not occur in gabbros and basalts because the Ti in these rocks goes to titanoaugite and ilmenite, respectively. It is a common mineral in ijolites.
In metamorphic rocks, it is more frequent in rocks such as mica-schists, amphibolites, glaucophane-schists, gneisses and marbles. It can occur in metasomatic rocks such as skarns and phenites.
It is often found in sedimentary rocks such as sandstones (graywackes) as a heavy mineral.
3. Mineral Associations
In plutonic igneous rocks it is associated with rock-forming minerals such as quartz, plagioclase (albite), alkali feldspars (orthoclase, microcline), micas (biotite, muscovite, chlorite, sericite) and amphiboles (green and brown hornblende).
Furthermore, it is associated with accessory minerals such as epidote, zircon, apatite, allanite, monazite, magnetite, ilmenite, pyrite, chalcopyrite and pyrrhotite.
When in marbles, it is associated with diopside, calcite and clinochlore (chlorite).
In low-silica rocks occurs with nepheline and sanidine.
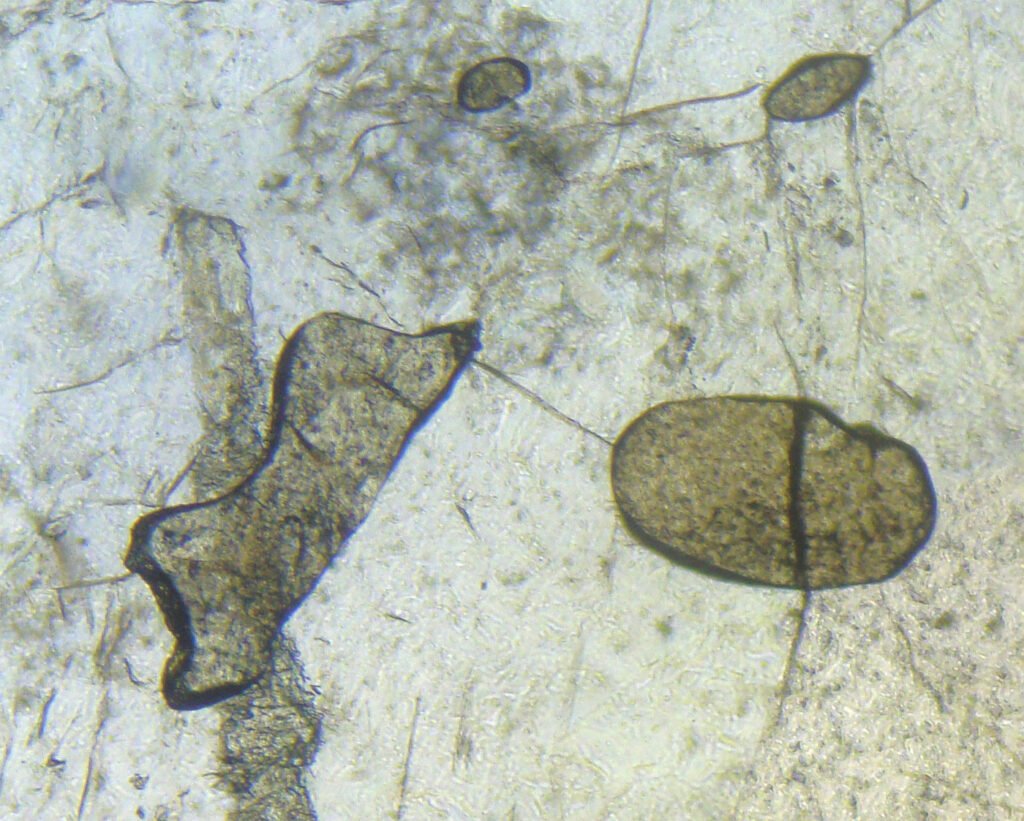
4. Transmitted Light Microscopy
Refraction indices: nα: 1.843 – 1.950 nβ: 1.870 – 2.034 nγ: 1.943 – 2.110
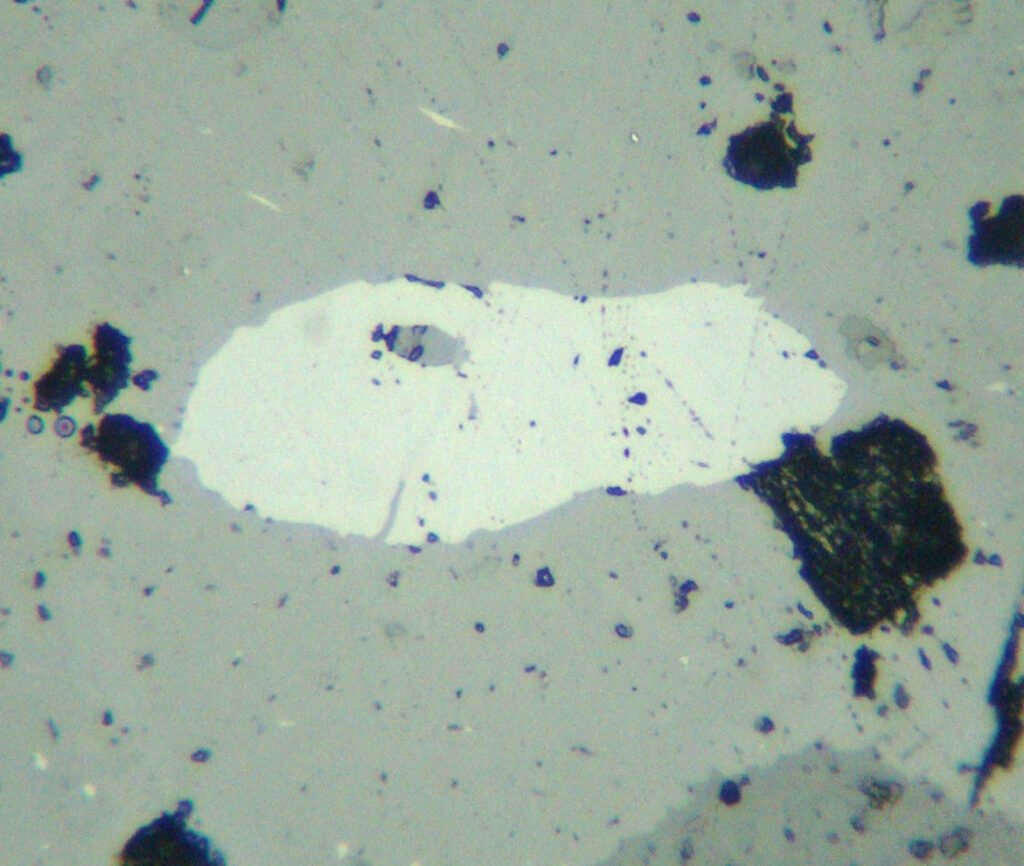
PLANE POLARIZED LIGHT – PPL
Color / Pleochroism: Brown, gray-brown to yellow-brown, with absent or weak pleochroism:
X = almost colorless,
Y = pale yellow,
Z = brownish.
It is necessary to pay close attention during the rotating of the stage to verify the pleochroism of the tianite.
Relief: Very high to extreme.
Cleavage: {110} good. It is a prismatic cleavage with two directions that, however, is generally not visible or poorly defined. Titanite normally has many fractures. It has a partition paralell to the twinning that can be more prominent than the cleavage.
Habits: Usually anhedral, completely xenomorphic. It only occasionally shows basal sections with rhombic outlines or diamond-shaped (wedge).
In metamorphic rocks, titanite tends to form clusters of small round grains.
When it occurs as inclusions in biotite, chlorite or hornblende and contain higher levels of Th, it generates black halos around it.
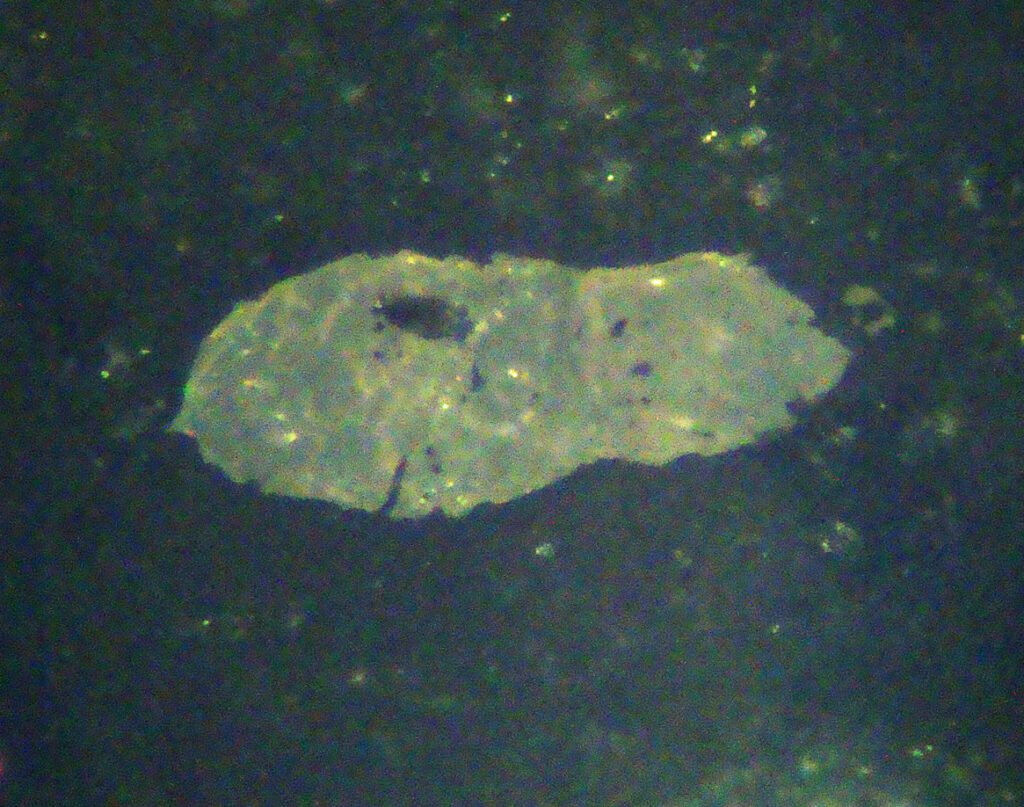
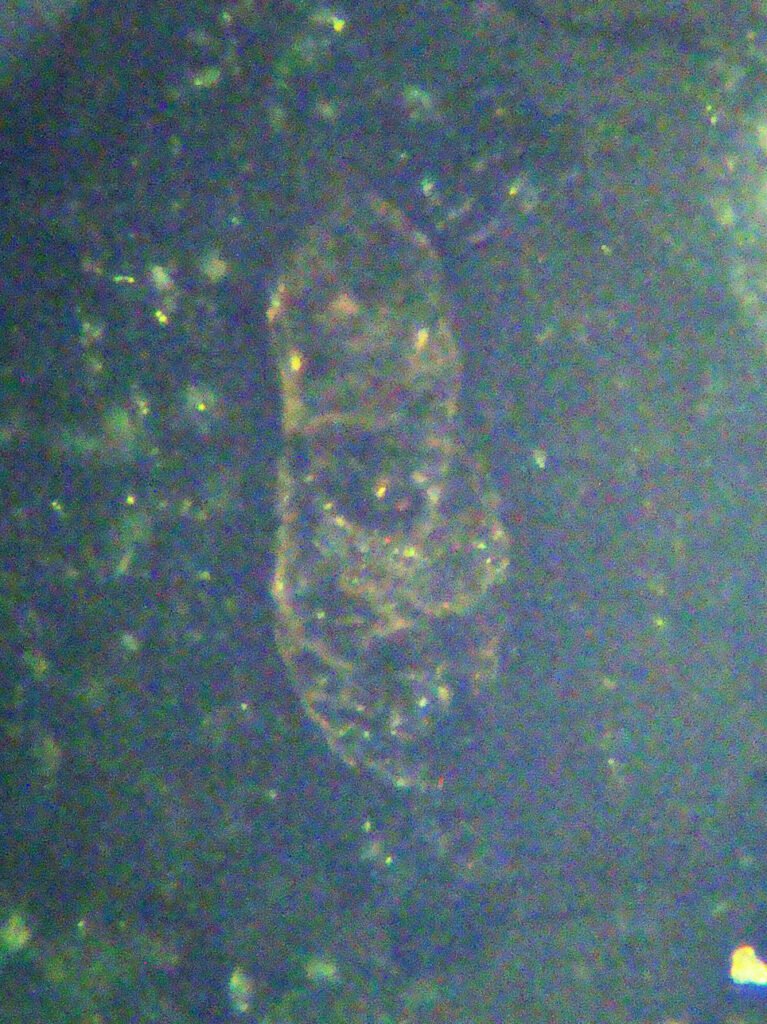
CROSSED POLARIZED LIGHT – XPL
Birefringence and Interference Colors: Birefringence from 0.1 to 0.192, very high, resulting in a high order white color, a very typical cream color. But the original strong brown color of the mineral masks this high interference color.
Generally the color of titanite does not change much from PPL to CPL. Some grains are very colored in CPL. Low birefringence grains often do not extinguish properly and show anomalous dark orange to dark blue colors.
Extinction: Oblique from 36 to 45º; symmetrical in basal (rhombic) sections.
Due to the high dispersion, some grains do not completely extinguish.
Elongation sign: Does not apply as titanite may be elongated in several directions.
Twins: Contact and penetration twins by {110} are common. Lamellar twins according to {221} occur more rarely.
Zoning: It can be zoned, in this case the margins have a higher refractive index.
CONVERGENT LIGHT
Character: B(+), hard to get.
2V angle: 17 a 56º
Alterations: under hydrothermal conditions it decomposes to leucoxene, a very fine-grained white to pale yellow mixture composed of rutile, quartz, calcite, apatite, ilmenite and minerals with Rare Earth Elements (allanite, monazite and bastnäsite). Leucoxene can be easily recognized using the Oblique Light technique.
Completely altered titanite grains become opaque due to the presence of ilmenite.
May be confused with: several other minerals.
Epidote has a lower relief, only one cleavage, and a much lighter color.
Cassiterite, zircon, rutile and xenotime are uniaxial.
Anatase and carbonates are U(-).
In very fine grains, titanite can be confused with epidote and allanite.















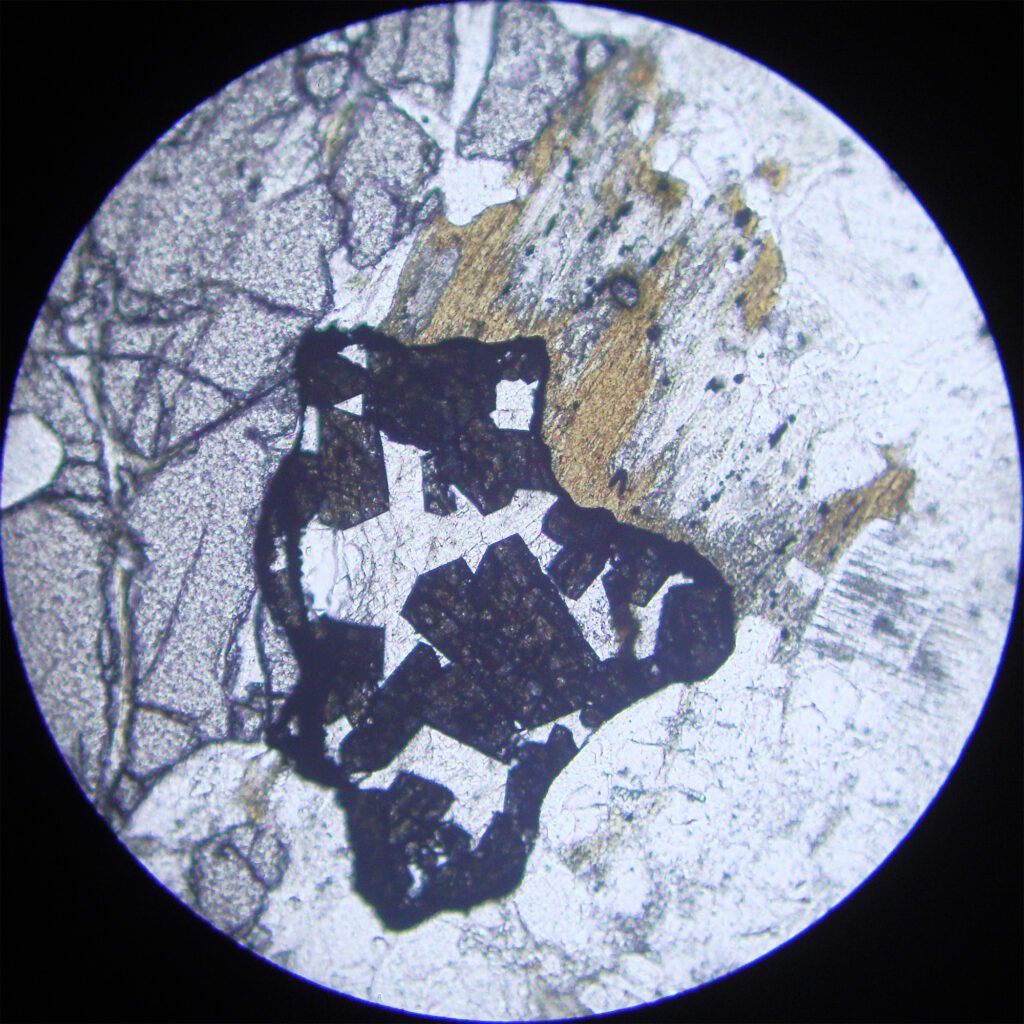

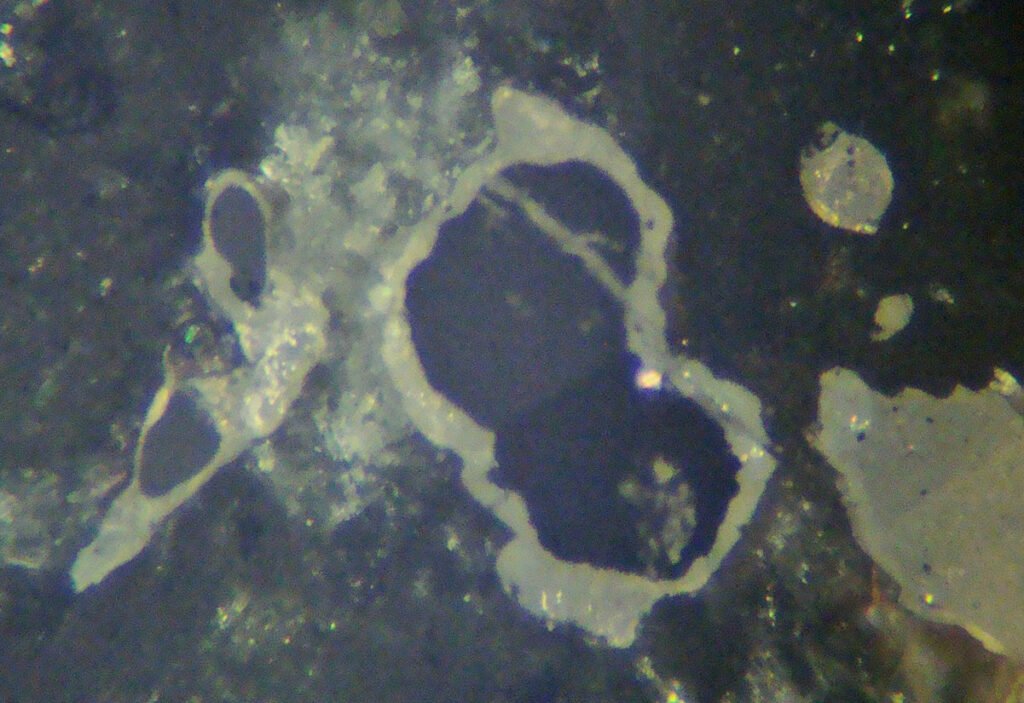
5. Reflected Light Microscopy
Reflected light microscopy is not the recommended analytical method for the identification of titanite. However, identification of titanite is quite simple in this technique. In addition, it is important to make a polished thin section or section to identify the opaque minerals that occur associated with titanite, such as ilmenite, magnetite, pyrite, chalcopyrite, pyrrhotite and others.
Sample preparation: titanite is very easily polished. Its polish is generally of better quality than other silicates that accompany it, such as quartz, feldspar, etc.
PLANE POLARIZED LIGHT – PPL
Reflection color: Light gray, much lighter than other silicates such as quartz, feldspars in general, amphiboles and pyroxenes.
Pleochroism: No.
Reflectivity: 9.43 – 11%, much lighter than other common gangue silicates!
Bireflectance: No.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: In basal sections there is no anisotropy or is masked by internal reflections, but in other sections the anisotropy is very strong, from light gray to dark gray.
Internal reflections: Generalized, abundant, yellowish to orange in color, can evolve to caramel and red. It resembles the internal reflections of large grain sphalerites and rutile.
When the color of titanite in hand specimen is greenish, the internal reflections also are green, a much rarer situation.
May be confused with: sphalerite and rutile.
General Characteristics:
Twins sometimes can be observed.
Pseudomorphoses of titanite on ilmenite or of titanite on the network of unmixed ilmenites of titanomagnetites can have an excellent development.
Substitutions of titanite for anatase (lighter in PPL) can occur, especially near deposits of metallic elements.
Extinction is symmetric in the basal rhombic sections.