
ANATASE
Anatase -TiO2 – is a relatively common oxide. It occurs as an accessory mineral in many igneous and metamorphic rocks and easily goes unnoticed. It does not constitute an ore.
Anatase is one of the five polymorphs of titanium oxide found in nature. The most common are brookite and rutile. Anatase may contain V, Nb, Sn, and Fe as impurities. There are four varieties of anatase.
1. Characteristics
Crystal system: Tetragonal, ditetragonal bipyramidal.
Color: Reddish brown, yellowish brown, yellowish, blue, gray to black. Rarely colorless.
Habit: Bipyramidal or tabular crystals, also cryptocrystalline masses. Granular.
Cleavage: [001] and [011] perfect.
Tenacity: Brittle.
Twinning: Rare by {112}.
Fracture: Subconchoidal.
Mohs Hardness: 5.5 – 6
Parting: No.
Streak: White, faintly yellow.
Lustre: Adamantine, metallic, specular.
Diaphaneity: Transparent.
Density (g/cm³): 3.79 – 3.97
2. Geology and Deposits
Anatase is a common accessory mineral in igneous and metamorphic rocks, also occurring in pegmatites. It can occur in Alpine-type tectonic rifts, formed from titaniferous minerals (ilmenite) in the host rocks such as schists and gneisses. It occurs in carbonatites, alkaline rocks, and some metasomatic rocks (adinole, a type of albitite). It may be present in epithermal metalliferous veins and in the host rocks thereof.
Anatase is the rarest of the TiO2 polymorphs. It can replace perovskite and very often occurs as cryptocrystalline to collomorphic masses when it replaces ilmenite, as one of the components of leucoxene (submicroscopic association of anatase with rutile and others).
As it is a fairly resistant mineral, it is found in placers (alluvial deposits), where it can form euhedral crystals in heavy sands containing ilmenite.
It is often secondary, formed from pre-existing titaniferous minerals. It forms as a product of the alteration of titaniferous minerals in weathered rocks. It can be diagenetic in sediments.
3. Mineral Associations
It is associated with many common minerals, such as quartz, plagioclase, K-feldspar (adularia), chlorite, titanite, pyrite, muscovite, fluorite, and calcite.
It also occurs with other oxides such as hematite, ilmenite, magnetite-Ti, perovskite, pyrochlore, brookite, and rutile.
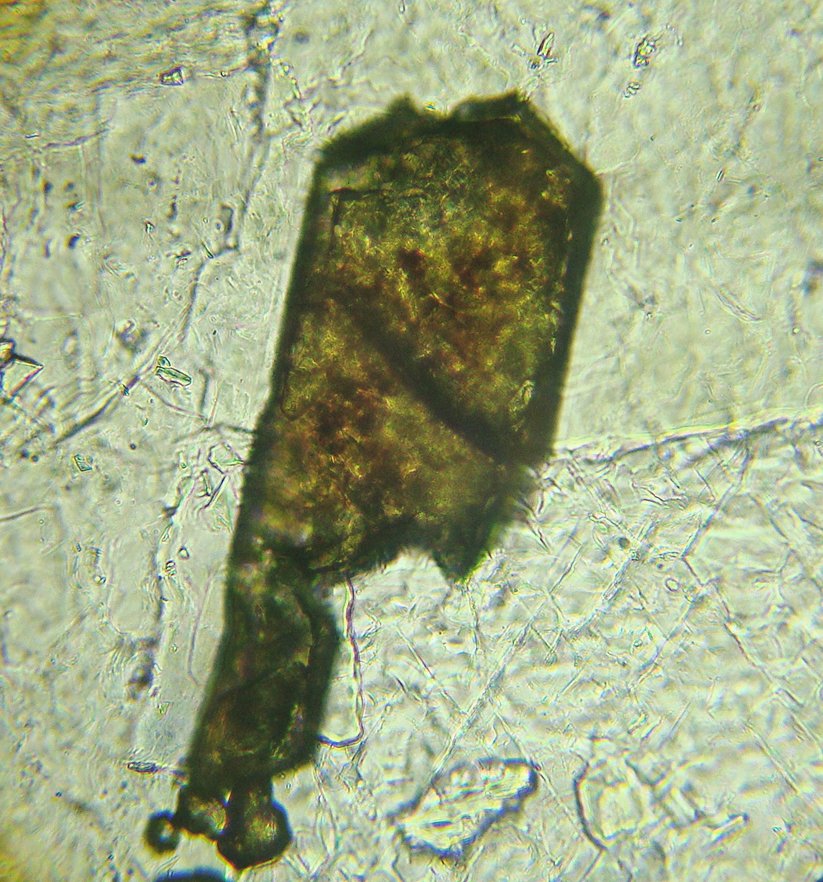
4. Transmitted Light Microscopy
Refraction indices: nω: 2,561 nε: 2,488
PLANE POLARIZED LIGHT – PPL
Color / Pleochroism: Quite colorful (can be yellow, yellow-brown, brown, dark blue, pale green, or black), with weak pleochroism that is sometimes very difficult to perceive. The pleochroism becomes stronger if the colors are more intense. The color distribution is usually uneven (spots) or zoned.
Deep-colored crystals can be mistaken for opaque crystals in thin section if a condenser is not being used. Therefore, it is useful to check the “opaque” crystals with the condenser placed in the path of the light to see if one of these “opaque” crystals is not anatase or another transparent mineral with dark colors.
Relief: Very high.
Cleavage: {001} and {011} distinct, difficult to see when the crystals are very small, which is very common.
Habits: Typically found in very elongated, pointed bipyramids, which gave rise to its name. “Anatasis” in Greek means “extensive,” “elongated.” Other possible habits are fine tabular and prismatic.
In thin section it is usually granular and very small.
Since rutile and anatase can occur together in the same rock, some care is needed to recognize and distinguish the two.
CROSSED POLARIZED LIGHT – XPL
Birefringence and Interference Colors: Maximum birefringence of 0.073: strong, intense colors, up to the 3rd order. The strong colors inherent to the mineral generally prevent, or mask, the detection of these interference colors.
Extinction: Tends to be parallel.
Elongation sign: SE(-), it is difficult to determine a function of the mineral’s strong inherent colors.
Twins: Rare.
Zoning: No information available.
CONVERGENT LIGHT
Character: U(-), can be anomalously biaxial in deep-colored crystals. In intensely colored crystals, obtaining the figure can be difficult or impossible.
2V angle: Very small if biaxial anomalous.
Alterations: It can alter to leucoxene, as can other minerals containing titanium.
May be confused with: Several other minerals.
Zircon and titanite are uniaxial and biaxial positive and generally have other habits and optical characteristics.
Perovskite exhibits characteristic twinning.
Spinel is isotropic.
Sphalerite is very similar, but is isotropic, which can be difficult to see.
Rutile can also be very similar, but is U(+), a finding that is generally impossible due to the minute size of the crystals.
But using the “Oblique Light” technique, minerals with titanium are easy to recognize, including its alteration product, leucoxene.



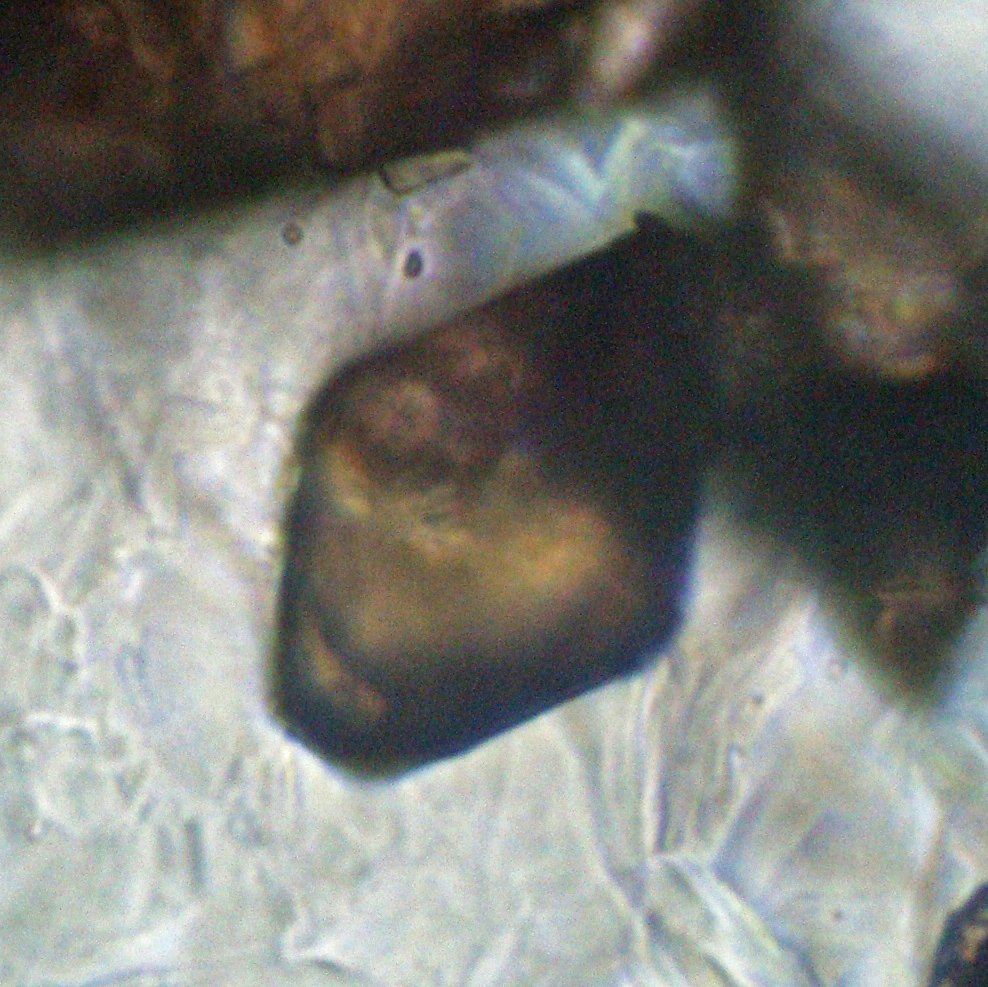
5. Reflected Light Microscopy
Reflected light microscopy is clearly not the recommended analytical method for identifying anatase. However, it is important to prepare a polished slide or section to identify the opaque minerals that occur associated with anatase, such as hematite, ilmenite, magnetite, and pyrite.
Sample preparation: Polishing anatase presents no problems, just as polishing rutile and brookite does not.
PLANE POLARIZED LIGHT – PPL
Reflection color: Bluish gray, a color very similar to that of rutile and sphalerite.
Pleochroism: Very faint, practically imperceptible.
Reflectivity: Around 20%
Bireflectance: No.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: It is anisotropic, but internal reflections mask the anisotropy.
Internal reflections: Light white or, more rarely, bluish to grayish-blue.
May be confused with: Anatase is much darker than rutile, does not exhibit pleochroism in its reflection, and has a different color. It is very similar to and easily confused with sphalerite, but sphalerite is usually associated with sulfides and exhibits internal reflections of other colors. Sections with very good polishing and concentrated sands make it very difficult to identify anatase.
General Characteristics:
Grain shape: Anatase occurs in small grains or as bipyramids.
Ilmenite substitutions by anatase can occur, including the formation of pseudomorphs. An intermediate product that can form during this process is titanite.
Fusiform demixing of ilmenite into anatase is possible.
Oriented intergrowths of anatase, rutile, and brookite occur.
Transformation of anatase into rutile can occur.
