CHALCOCITE
Chalcocite – Cu2S – is a relatively common sulfide, being one of the most profitable Cu ores, with almost 80% by weight Cu. Crystals are very rare and obtain high prices in the market of collectible minerals.
The planetary abundance of copper and sulfur, its wide distribution in the most varied geological environments and the high reactivity of the two elements generated almost 300 different mineral species that contain them. There are nine different minerals formed only by Cu + S:
Copper sulfide formed at low temperatures (T<~105ºC) is monoclinic chalcocite.
At high temperatures (T>~105ºC) (in blast furnaces too) digenite (Cu9S5 trigonal) is formed.
Another copper sulfide, djurleite (monoclinic Cu31S16), is easily confused with chalcocite.
To add to the confusion, another copper sulfide, orthorhombic anilite (Cu7S4), transforms into digenite during the making of the polished section, by polishing. Other copper sulphides are roxbyite (monoclinic Cu9S5), spionkopite (trigonal Cu39S28), yarrowite (trigonal Cu9S8), geerite (trigonal Cu8S5) and covellite (hexagonal CuS).
The situation becomes more difficult because in several of these minerals there are substitutions of Cu by varying contents of Ag, Fe and Mn, in addition to substitutions of S by Se and Te. In addition, there are crystals with unmixings of digenite x chalcocite and digenite x covellite. And there are serious sample preparation problems, which are discussed further below. This set of complicators has historically caused much confusion and misidentification (More: Pósfai, M. & Buseck, P.R. (1994): Djurleite, digenite, and chalcocite: Intergrowths and transformations. American Mineralogist, 79, 308-315).
Chalcocite is classified in the Chalcocite-Yarrowite Series, in the Chalcocite-Digenite Group, forming a series with yarrowite. It has four varieties: a perimorphosis on pyrite, a pseudomorphosis on galena, a paramorphosis on high-temperature chalcocite and an Ag-containing variety.
Crystals reach sizes of 12 cm when short prismatic and 25 cm when long prismatic. Twins on {110} are common and macroscopically produce pseudohexagonal stellate shapes; microscopically, they appear as parallel lamellae. The fusibility of chalcocite is 2 – 2.5.
There are 3 types of chalcocites with different habits, formed in different environments and presented below in increasing order of abundance.
a) Chalcocite formed at high temperatures (>~103ºC) by the transformation and recrystallization of digenite.
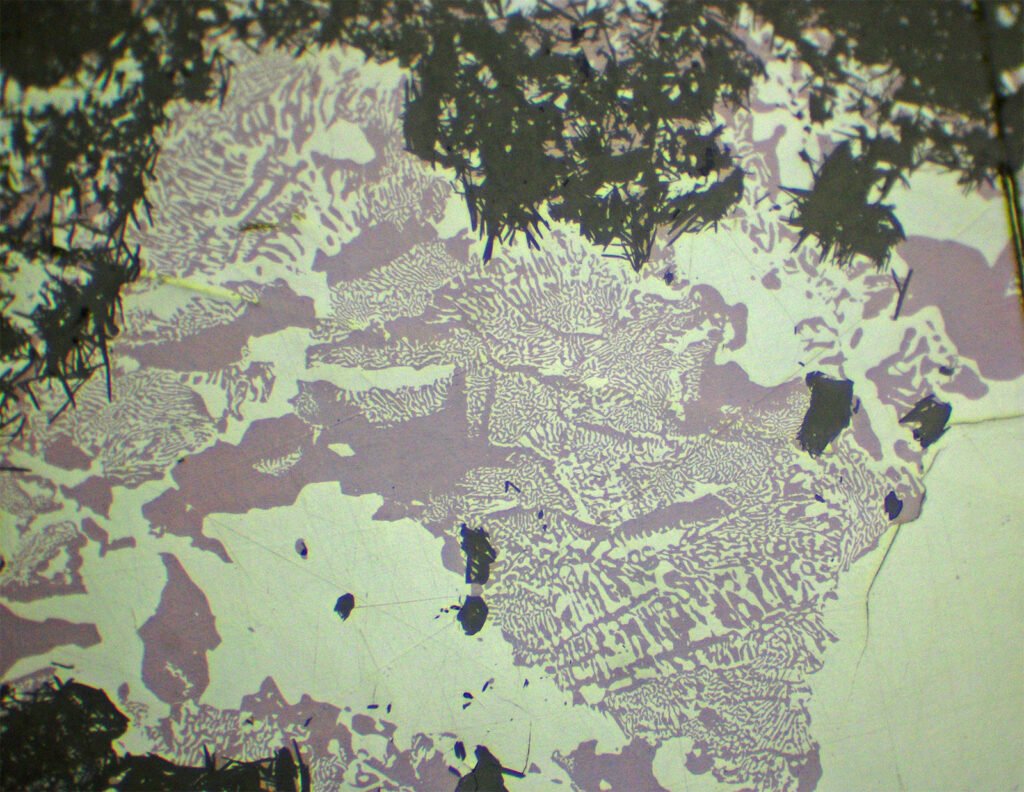
The recrystallization and transformation of the original copper sulphide (digenite) generates mixtures of chalcocite + digenite or chalcocite + covellite or chalcocite + bornite. In all cases an excellent pseudo-octahedral structure of chalcocite lamellae appears with (001) parallel to (111) of the digenite. They have been often described as “lamellar chalcocites” (= “Lamellarer Kupferglanz”) and should not be confused with the original digenite lamellae. The spaces between the lamellae, with a thickness of 0.03 mm, can be occupied by bornite, digenite itself, digenite + chalcocite or covellite, etc. Viewed in CPL, they often evidence the previous trigonal structure.
In some cases, the infillings preserve the lamellar structure, preventing its recrystallization. In other cases this recrystallization has occurred and the only evidence of its existence is randomly dispersed very fine-grained chalcocite aggregates. Coarse-grained pseudomorphoses on bornite, digenite or pyrite are very common.
Very similar lamellar structures can be generated by replacing bornite with chalcocite lamellae parallel to (111). With some practice, it is observed that in chalcocite recrystallized from digenite the proportion of chalcocite and bornite is approximately equal, while in cases of replacement of bornite by chalcocite the proportions are very varied.
But lamellar structures in some cases are not present, even in chalcocites clearly formed from digenites. There is a “pink-gray chalcocite” formed by the demixing of crystals with Cu2S + Cu5FeS4. The approach of these cases is beyond the scope of this approach.
b) Chalcocite formed at low (T<~105ºC) temperatures directly from the reduction of copper sulfate solutions in the cementation zone.
These chalcocites present very varied structures. Often the grains are coarse with sharp cleavage, or there are inclusions of thin covellite plates parallel to (001). These chalcocites can also occur in red beds, sedimentary basins and other types of geological environments.
c) Chalcocite formed at low (T<~105ºC) temperatures directly from sulfides (chalcopyrite, pyrite, sphalerite, galena and many others) in the cementation zone.
These supergenic chalcocites are economically the most important chalcocites. They usually form compact masses of homogeneously fine grains, not very intertwined with each other, known as “steely chalcocite”. Another type, “sooty chalcocite” is usually formed by a mixture of very fine-grained supergenic black chalcocite mixed with supergenic digenite and covellite, which simulates isotropy.
Chalcopyrites in the lower portion of the iron cap or in the upper part of the cementation zone often present this “sooty chalcocite” in the form of thin surface films, flakes and caps.
Macroscopically, this type of chalcocite has a conchoidal fracture and, in a fresh(!) fracture, has a light blue-white color. By corrosion, even by natural oxidation processes, the polygonal structure of the grains (“crackled porcelaine”) stands out very well. There are abundant replacement structures and relics of the replaced mineral. In some cases there are rhythmic structures (bands) indicative of colloidal deposition. In other cases, recrystallization has largely erased the structures.
1. Characteristics
Crystal system: Monoclinic prismatic.
Color: Black lead gray, black-grey, black-blue. Quickly tarnishes to brown colors. Sometimes iridescent.
Habit: Massive, granular. Rare short prismatic or pseudo-orthorhombic tabular crystals (twins). Powdery.
Cleavage: {110} indistinct. Striations// to [100], common and deep.
Tenacity: Brittle, somewhat sectile.
Twinning: See above.
Fracture: Conchoidal.
Mohs Hardness: 2.5 – 3
Parting: No.
Streak: Lead-gray black.
Lustre: Metallic.
Diaphaneity: Opaque.
Density (g/cm³): 5.5 – 5.8
2. Geology and Deposits
Chalcocite is usually secondary (supergenic), formed in the oxidation zone of copper ores, which can be hydrothermal veins or large bodies of low-grade “porphyry copper” ore, due to copper leaching by alteration of the original minerals. In arid climates, chalcocite forms an approximately horizontal level within or below the oxidation zone that corresponds to the position (past or present) of the phreatic aquifer. This level is called the “chalcocite blanket” and is the most copper-rich horizon of the entire occurrence.
Chalcocite is sometimes found as a primary mineral in hydrothermal veins.
It rarely occurs in basic igneous rocks (basalts) and can form in volcanic fumaroles.
It can occur in contact metamorphic deposits (scarnites) and in pegmatites.
It is found in “red beds” Cu deposits, hosted in sandstones.
3. Mineral Associations
Chalcocite occur associated with common gangue minerals such as quartz and calcite.
It naturally occurs with many other Cu minerals, both primary (bornite, chalcopyrite, djurleite) and secondary (native copper, covellite, chrysocolla, malachite, azurite, digenite, enargite, tenorite, luzonite, delafosite, stromeyerite, atacamite, etc.).
In the oxidation zones, Fe oxides and hydroxides, such as goethite, also are associated.
It occurs with other sulfides such as pyrite, pyrrhotite, sphalerite, galena and molybdenite.
It is found associated with silver, stannite, wittichenite, tellurides and selenides.
4. Transmitted Light Microscopy
Not applicable, as chalcocite is completely opaque.
5. Reflected Light Microscopy
Sample preparation: Chalcocite is relatively easy to polish, producing a high gloss surface. The polishing hardness is much higher than the hardness of acanthite, approximately equal to the hardness of digenite and galena and lower than the hardness of bornite and tetrahedrite. In relation to covellite, the hardness of chalcocite is greater than the hardness of the basal covellite sections and less than the hardness of the prismatic covellite sections.
Usually the surface is heavily scratched, especially when there are small (hard) grains of pyrite embedded in the chalcocite. Careful polishing is required and long polishing should be avoided because they produce too high relief in the associated minerals and because the chalcocite surface is smudged due to the exaggerated surface film. To avoid this chalcocite surface blurring, it should never be polished dry or under high pressure. Too much pressure produces surface films on the chalcocite that later darken to blue colors. In addition, polishing grooves produced with coarse abrasives (which should be avoided in chalcocite ores) and which were not eliminated during polishing, are filled with mineral paste during the polishing process, appear bluer than the correct reflection color. and can generate pseudo-structures.
Poor quality polishing make the chalcocite reflection color appear more bluish than normal. After polishing, the section must be dried immediately and very carefully. Incorrect drying results in surfaces with abnormal blue colors. The section cannot be impregnated at high temperatures (T>60º) nor should it be observed under a microscope with oil immersion for long periods (except for microscopes with LED cold light), because the structure of the chalcocite begins to change and associated covellites are reabsorbed. The resin in the polished section must not contain sulfur, as this reacts with the chalcocite to produce a mold-like film.
The surfaces of the polished sections (and hand samples) of the collections change slightly due to the formation of a brown crust of CuS. Leaving the sections exposed to the humid atmosphere, an air corrosion (turbidity) develops on their surfaces, which shows possible existing structures in the chalcocite.
PLANE POLARIZED LIGHT – PPL
Reflection color: Gray-white to bluish-white. Colors with abnormal greenish hues may occur.
Compared to the color of galena, the color of chalcocite is distinctly bluish.
Compared with the color of pyrite, the color of chalcocite is bluish gray.
Compared to the color of covellite, the color of chalcocite is white, without a pinkish tint.
Compared to the color of bornite, the color of chalcocite is bluish white.
Compared to the color of native copper, the color of chalcocite is bluish gray.
Pleochroism: Extremely weak, almost impossible to notice.
Reflectivity: 29.93%
Bireflectance: No.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: Weak to distinct anisotropy, between emerald green and light pink, difficult to observe with exactly crossed nicols.
It must be observed uncrossing the nicols in 2o and even then it is sometimes difficult to observe. With a strong LED light, observation is easier.
Apparent isotropy can arise when too much pressure is used during the polishing of the sample.
Internal reflections: No.
May be confused with: other white minerals, isotropic or with weak anisotropy, such as galena, tetrahedrite-tennantite and others.
General Characteristics:
Cleavage (111) may be visible, but only in larger crystals.
Polishing scratches may occur in great number.
Twinning according to (111) can sometimes be seen, with hexagonal shapes.
Replacement structures of other minerals by chalcocite are very frequent, both replacement by ascending (primary) and descending (secondary) chalcocite. The textures generated by the primary and secondary chalcocites can be identical. Descending chalcocite can replace almost all sulphide ores, especially pyrite, chalcopyrite, sphalerite, enargite, stannite, bismuthinite, tennantite-tetrahedrite, bornite, galena and many others. In rare cases, it replaces oxidized carbonates and ores. When chalcocite meets chalcopyrite, bornite reaction edges often form. Magnetite, on the other hand, has never been observed in contact with chalcocite. The replacement of minerals by chalcocite normally follows crystallographic directions, with the newly formed chalcocite forming lamellae // (010) in enargite or // (100) in bornite, for example. Conversely, primary chalcocite can be replaced by chalcopyrite and bornite at all stages. Secondary chalcocite is replaced by acanthite, native silver and some other silver minerals, cuprite, native copper, covellite and oxidized copper ores such as malachite and others. Replacement structures can be found in all copper deposits, at any level (iron hat, oxidation zone, cementation zone). Replacement can often eliminate any trace of the primary ore.
Oriented intergrowths are common and are described above.
Rhythmic depositions in the form of stalagtite masses are rarer, but locally frequent.
Myrmekitic intergrowths are very common, especially with bornite, but also with galena, clausthalite, covellite, hematite, wittichenite, stromeyerite and others. Myrmekites with bornite are very frequent, ranging from macroscopic to submicroscopic scales.
Pseudomorphoses of chalcocite can be on bornite, covellite, chalcopyrite, pyrite, enargite, millerite, galena and sphalerite.