CHROMITE
Chromite – FeCr2O4 – is a relatively common oxide and constitutes the most important ore of chromium.
The name “chromite” is commonly used to designate any Cr-rich mineral of the Spinel Group, especially for the chromite-magnesiochromite series. As pure chromite is extremely rare, the “chromites” in the literature are actually magnesiochromites, which is another mineral species. Chromite apparently forms a complete solid solution series with many other members of the group, in the chromite-hercynite, chromite-spinel, chromite-magnetite, and chromite-magnesiochromite series.
Chromite is the Fe analogue of zincchromite, cochromite, manganochromite and magnesiochromite and is the Cr analogue of hercynite, coulsonite and magnetite. It is dimorphic with shieite; contains Mn, Zn, Ti and others. Chromite crystals tend to form round-edged octahedrons. It may be weakly magnetic.
1. Characteristics
Crystal system: Cubic hexaoctahedral.
Color: Black to black-brown. May have shades of green.
Habit: Massive, compact, fine-grained, globular, rarely octahedral. Crystals up to 1 cm.
Cleavage: No.
Tenacity: Extremely brittle.
Twinning: On {111}, Spinel Law.
Fracture: Irregular.
Mohs Hardness: 5.5
Parting: Possible, on {111}.
Streak: Tan to brown.
Lustre: Submetallic to metallic, may be resinous.
Diaphaneity: Opaque.
Density (g/cm³): 4.5 – 4.8. Reduce to 4.2 if high in Mg and Al.
2. Geology and Deposits
The primary chromite deposits are stratified ultramafic intrusions, formed by intrusive bodies of large proportions where successive levels of chromite, olivine, pyroxenes and plagioclases crystallize in varying proportions during cooling. In these occurrences it is so abundant that it forms the rock chromitite, which contains 90% or more of chromite. A stratified body of this type is the Bushveld Complex in South Africa. This type of occurrence is called the “Stratiform Type”.
The most important secondary deposit forms when these stratified intrusive bodies integrate orogenetic processes. They are sheared, folded and metamorphosed to varying degrees, now constituting rocks such as peridotites (dunites), serpentinites, limburgites, pyroxenites, picrites, gabbros and norites. This type of occurrence is called “Alpine Type”.
Chromite can occur detrital in placers and laterites. It is common in almost all types of meteorites.
3. Mineral Associations
Chromite occurs with basic rock-forming minerals, such as clinopyroxene (titanoaugite), plagioclase, olivine, ilmenite and magnetite. It occurs with associated magmatic sulfides such as pyrite, pentlandite, pyrrhotite, chalcopyrite, heazlewoodite, and millerite.
In addition, it is associated with rutile, minerals from the Platinum Group (including native platinum), hematite, serpentine, enstatite, chlorite, talc, garnet (uvarovite), ulvöspinel, corundum, pyrolusite, cryptomelane, graphite and skolaite.
4. Transmitted Light Microscopy
Refraction indices: n: 2.05 – 2.16
PLANE POLARIZED LIGHT – PPL
Color / Pleochroism: Dark brown to almost opaque. In very thin plates and at grain boundaries, chromite may be translucent with dark brown to reddish-brown color. This is generally not possible to observe, even in those portions of the thin section that were left with thicknesses below 30 microns.
Relief: High relief, may show dark edges around grains.
Cleavage: Does not present. Can show partition.
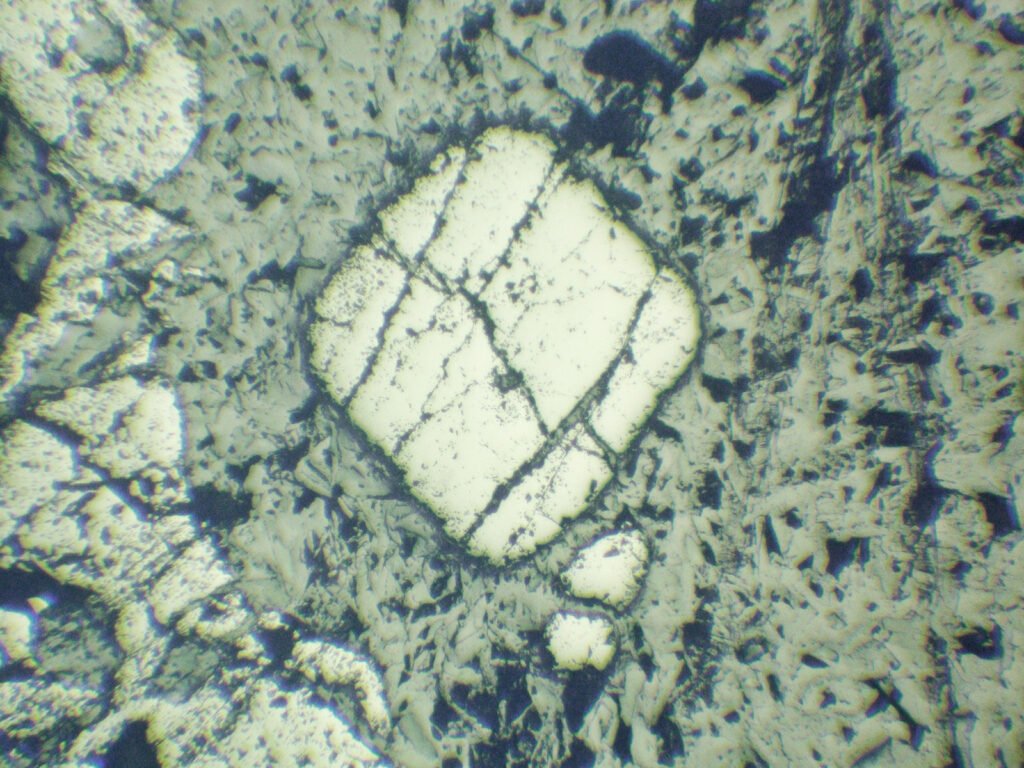
Habits: It is usually granular anhedral, may be very fragmented, sheared. It can form octahedral crystals, in which case it can have triangular, trapezoidal, square or diamond-shaped (rhombic) sections.
CROSSED POLARIZED LIGHT – XPL
Birefringence and Interference Colors: Isotropic.
Extinction: Isotropic.
Elongation sign: Isotropic.
Twins: Isotropic.
Zoning: Isotropic.
CONVERGENT LIGHT
Character: Isotropic.
2V angle: Isotropic.
Alterations: generally shows no alteration, but may alter to goethite (“limonite”) and other Fe, Mg, and Cr oxides and hydroxides, as well as to Cr-containing clay minerals.
May be confused with: other opaque minerals common in basic rocks, such as magnetite and ilmenite. Identification of chromite is difficult under Transmitted Light and must be obtained through Reflected Light.


5. Reflected Light Microscopy
Sample preparation: Due to its high hardness, comparable to that of pyrite and magnetite, chromite acquires a good polish, but only very slowly. It is a little less hard than hematite. Literature insists on the difficulty of obtaining this good polish. Therefore, it is concluded that a low quality polish is still a diagnostic aspect for chromite.
PLANE POLARIZED LIGHT – PPL
Reflection color: White gray with brown tones; shades vary with changes in chemical composition.
Compared with the color of magnetite, the color of chromite is much darker.
Compared with the color of sphalerite, the color of chromite is similar, a little darker.
Compared with the color of ilmenite, the color of chromite is much darker.
Pleochroism: No.
Reflectivity: Low (11.76%)
Bireflectance: No.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: Isotropic. May have a weak anomalous anisotropy if with zinc. This occurs especially in zoned crystals or in crystals whose structure has been deformed by tectonism.
Internal reflections: Occasional red-brown in the thinner portions and in chromites with Mg and Al. Chromites with high iron content do not show reflections. Reflections are usually absent.
May be confused with: several other minerals, including some dark colored silicates. Chromite does not have any easily recognizable diagnostic properties.
Magnetite is very similar and occurs in the same paragenesis. Its reflectivity is higher.
Ilmenite in PPL has a similar color but is anisotropic.
General Characteristics:
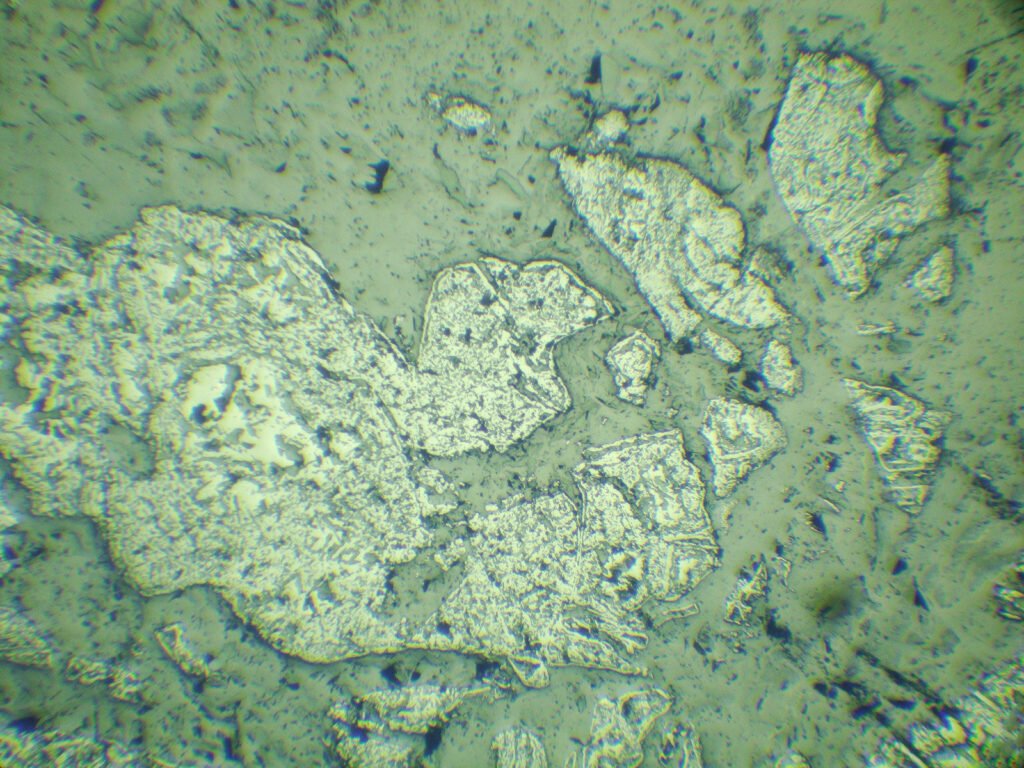
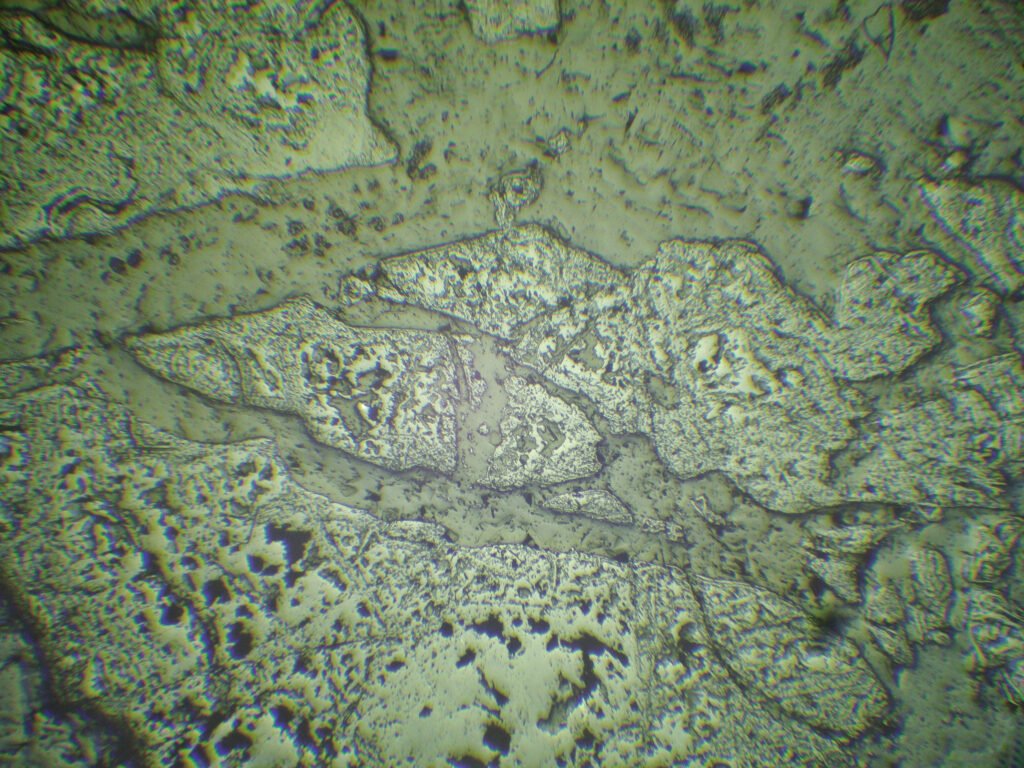
Grain shape: generally forms coarse granular aggregates with an uneven grain size, whose grains may be very fractured. It may have octahedral or cubic outlines, often rounded. Crystals are simple, with few combined shapes.
Cleavage does not shows, but sometimes the partition is distinct.
Cataclasis is very frequent, caused by tectonism or due to serpentinization. It can be very pronounced, with no evidence of cleavage, twin lamellae by pressure or recrystallization. In some cases the cataclasis is extremely pronounced and there is no mineral filling the fractures, making the chromites powdery like powdered chocolate, which makes their use as refractories difficult due to the “bursting” they present (they explode under dilatation).
Zonation is not uncommon, with crystals showing darker nuclei. In rare cases, the edges are darker. In zoned crystals, the edge is richer in Fe and Cr and is more reflective (lighter), while the core is richer in Mg and Al. Care must be taken not to confuse zonation with edge color changes caused by alteration.
Twins do not occur.
Partition is rarely visible, but it can be sharp. Inclusions and exsolutions arranged in parallel lines often simulate cleavage.
Substitutions are rare due to the chemical stability of chromite. If the host rock is completely serpentinized, chromites with corroded edges can occur. In this case, there are substitutions for serpentine. In some epi- and mesometamorphic ultrabasic rocks there are magnetite porphyroblasts that contain in their center a chromite fragment that survived metamorphism and was oriented in a magnetite manner.
Intergrowths with other minerals are infrequent, but can occur, sometimes oriented, with hematite, ilmenite, magnetite, rutile and ulvospinel.
Poikilitic textures generated by the intergrowth of chromite with olivine may occur.
Inclusions of gangue minerals as well-developed negative forms are frequent. Inclusions in the form of droplets of sulfides and silicates also occur. Radioactive inclusions can occur, with halos around them.
Unmixings are relatively rare. Unmixings of hematite and ilmenite occur. Hematite forms thin plates parallel to (111) or complete networks, which can constitute 20% of the mass. Very iron-rich chromites may show ilmenite demixes that disintegrate into hematite + ilmenite. But even iron-poor chromite can show ilmenite demixes. Rutile, a frequent accessory mineral, can form oriented crystals in chromite ((100) of rutile arranged parallel to (111) of chromite). Magnetite can form quasi-myrmekitic dismixtures (?) in chromite. Other “chromites” show dismixtures of magnetite + chromite, with chromite starting from intergranular films until it occupies the entire grain.
Magnetite crusts, which may be partially martitised, are common in many occurrences. Replacement of chromite by magnetite also occurs.
Porphyroblastic textures may occur, with porphyroblasts having a more iron-rich nucleus.
Alteration acts on chromites very slowly and normally cannot be noticed. Crystals of slightly different compositions show markedly different behavior in relation to alteration. Chromite can change to a mixture of at least three new chromium minerals.