COVELLITE
Covellite – CuS – also known as “covelline”, is a very common sulfide, an ore of Cu. It may be the main Cu ore mineral in the deposit, but is usually of minor importance, occurring in small amounts as alteration material.
Macroscopically it can be recognized by its deep blue, almost black color. However, it rarely forms larger crystals; it is usually massive and occurs in very small, microscopic lamellae. In most cases it is relatively pure, with only Cu and S. Its varieties are covellite-Ag and covellite-Se. It can also contain Pb and Fe and can pseudomorphically replace several other minerals. Crystals reach up to 10 cm.
1. Characteristics
Crystal system: Hexagonal, dihexagonal bipiramidal.
Color: Dark indigo blue to black, blurring heavily to purple, purplish and red.
Habit: Usually massive, granular and foliate. Lamellas are common. Forms hexagonal tabular crystals in rosettes.
Cleavage: {0001} perfect, {10-10} indistinct.
Tenacity: Flexible.
Twinning: No.
Fracture: Irregular.
Mohs Hardness: 1.5 – 2
Parting: No.
Streak: Gray to black, with metallic lustre.
Lustre: Submetallic to resinous, dull when massive.
Diaphaneity: Opaque. Very thin lamellae are translucent green.
Density (g/cm³): 4.6
2. Geology and Deposits
Covellite is formed as a secondary mineral in the oxidation zone of copper sulphide ores deposits, by the alteration of chalcopyrite, bornite, chalcocite and others. Hardly a Cu ore does not show covellite. Covellite can form a surface layer of tarnish on other minerals.
In porphyry copper deposits, covellite is a common product of supergenic alteration, participating in the assemblage of secondary minerals that develops there. In “red-bed” copper deposits, covellite is associated with chalcocite. Covellite is also found in IOCG (iron oxide copper gold) ore deposits, where it develops from chalcopyrite, which is the main primary copper mineral.
It rarely occurs as a primary mineral in high-temperature hydrothermal deposits. Very rarely occurs in volcanic sublimates, where it has been recognized, so much so that the type locality is the Vesuvius volcano in Italy.
3. Mineral Associations
Covellite occur associated with many other copper minerals, primary or secondary. Among the primary minerals, the most important are bornite, chalcocite, chalcopyrite, djurleite, digenite, enargite, luzonite and tetrahedrite-tennantite.
Among the secondary ones, there are at least a dozen green minerals (malachite, pseudomalachite, dioptase, antlerite, atacamite, paratacamite, libethenite, chalcanthite, brochantite and chrysocolla), at least five blue minerals (azurite, plancheite, linarite, chrysocolla and chalcanthite), two red minerals (native copper and cuprite) and one black mineral (tenorite); several others are possible.
It is also associated with quartz, calcite, other sulfides (pyrite, marcasite, linnaeite, etc.), oxides (magnetite, hematite), native sulfur, native silver, goethite (limonite) and others.
4. Transmitted Light Microscopy
Under the Transmitted Light microscope, covellite is completely opaque, does not show the blue color, nor is it possible to perceive fine translucent green flakes, as reported in the literature.
Its refractive indices are nε = 2620 and nω = 1450. It has a maximum birefringence of 1.170 and extremely high relief. It is uniaxial positive.
5. Reflected Light Microscopy
Sample preparation: The polishing hardness is very low, a little lower than those of galena and chalcopyrite. It is similar to that of chalcocite: basal sections a little less hard than chalcocite and prismatic sections a little harder than chalcocite. The hardness is greater than that of acanthite and cerargirite. Despite this low hardness, covellite acquires an excellent polish with ease.
As with all low hardness minerals (graphite, molybdenite, etc.), during grinding and polishing, avoid pressing the sample too hard against the polishing cloth. It is also advisable to polish with a finer abrasive and with more water than normal, to reduce the amount of polishing scratches.
PLANE POLARIZED LIGHT – PPL
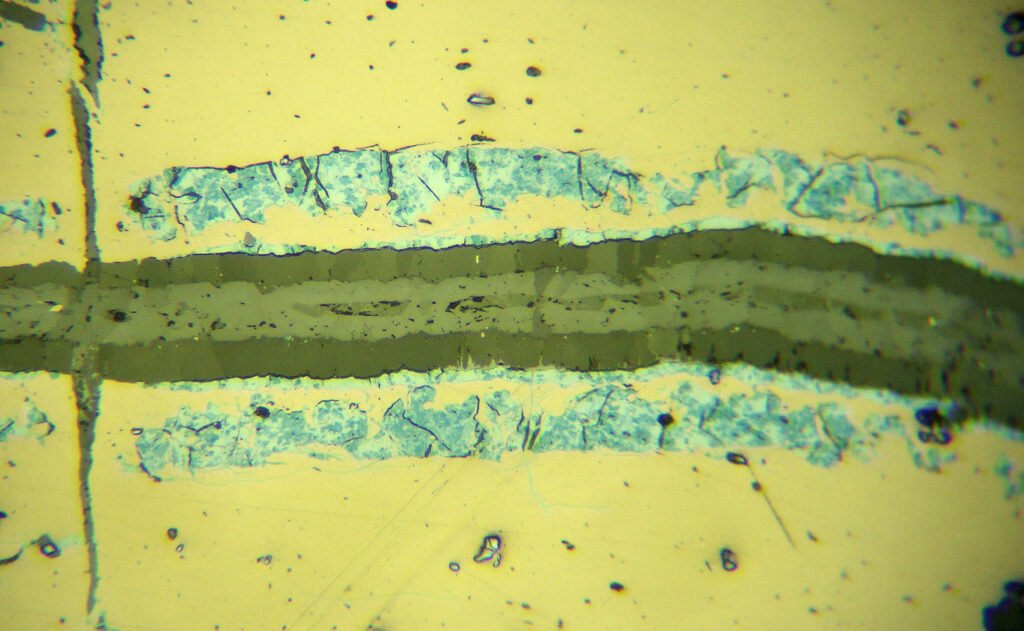
Reflection color: Deep blue to bluish-white; the shade of blue depends on the section in which the grain was sectioned.
Pleochroism: Strong between intense blue or soft violet with white-blue. It is the most notable and diagnostic pleochroism of all ore minerals! Basal sections show indigo blue colors and do not show pleochroism because they constitute isotropy sections.
Reflectivity: 3.4 – 19.1
Bireflectance: Strong.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: Extremely strong anisotropy in shades of intense bright orange to brown, less commonly reddish brown. Color effects of this intensity are known in very few ore minerals.
When the “covellites” present, in XPL, blue colors instead of orange or much weaker and less luminous orange tones, it is spionkopite (Cu39S28) and/or yarrowite (Cu9S8), a mixture that in ancient literature was designated as a variety of covellite and called “blaubleibender Covellin” (in German, “covellite that remains blue”).
Internal reflections: Does not present. When the section is very oblique to the cleavage, some reflections may occur, but they are just narrow colored stripes that do not show the green color that covellite presents in extremely fine flakes.
May be confused with: It is impossible to confuse covellite with any other mineral due to the outstanding optical characteristics of covellite.
General Characteristics:
Grain Shape: Covellite is normally idiomorphic, even being an alteration product that formed after the associated ore minerals. The crystals are lamellar, usually fine or very fine, micaceous. Crystal size varies widely (submicroscopic to centimeter) and may appear in radial aggregates. Rosettes and aggregates may occur.
Anomalous colors in covellite can be of two types. (i) There are covellites which show weaker blue colors and which are almost always arranged as small lamellae parallel to {0001} in chalcocite. They can occur together with “normal” covellites in the same section. (ii) Another case is of “covellites” that show, in CPL, the same colors as in PPL (gray-blue instead of the bright orange tones of “normal” covellites) or else much weaker and less luminous orange colors. It is spionkopite (Cu39S28) with yarrowite (Cu9S8), a mixture that in ancient literature was designated as a variety of covellite and called “blaubleibender Covellin” (in German, “covellite that remains blue”). They are alteration products that form from chalcocite or digenite. They can replace chalcopyrite, bornite or digenite.
Cleavage parallel to {0001} can only be visible if the crystals are large.
Polishing grooves, very fine, are often visible, especially on larger crystals and in CPL.
Twins do not occur.
Zonation does not occur.
Deformations are possible, but as covellite is a late-forming mineral, deformations due to tectonics are rarely visible. Deformed lamellae, on the other hand, occur.
Intergrowths, often oriented, occur with many other sulfides (sphalerite, chalcopyrite, chalcocite, digenite, bornite, galena, marcasite, pyrite, cuprite and aikinite). Many of these intergrowths can be textures formed by substitutions.
Substitutions are extremely common, as covellite is an oxidation product that occurs in the oxidation zone as an initial alteration phase or as a cementing mineral.
Substitutions 1: covellite replaces chalcopyrite and can form complete pseudomorphoses. This substitution can be (i) completely irregular in relation to the chalcopyrite forms or (ii) the covellite can have the cleavage (0001) arranged parallel to the faces of the main chalcopyrite sphenoid. (iii) Sometimes, the chalcopyrite is completely or almost completely replaced, only the arrangement of the covellite lamellae informs its origin from the chalcopyrite. (iv) In rare cases, the alteration of the chalcopyrite forms a mixture of covellite and Marcasite, which are intergrown in an oriented manner. (v) In subvolcanic deposits, chalcopyrite, with the addition of S, is replaced by pyrite and covellite.
Substitutions 2: Covellite can partially or fully replace (pseudomorphs) a number of minerals, including enargite, tennanthite-tetrahedrite, bornite, chalcocite, stromeyerite, emplectite, stannite, sphalerite, galena, and pyrite.
Substitutions 3: covellite selectively develops in unmixings with bornite. When bornite is unmixed into chalcopyrite, covellite develops much further into bornite. When bornite is unmixed (myrmekites) into chalcocite, covellite develops much further into chalcocite.
Substitutions 4: in ore minerals that do not contain copper, covellite may also occur, penetrating through the cleavage planes. This process was verified in sphalerite and galena. In sphalerite, covellite can occupy large portions. In galena, substitution by chalcocite may occur, which changes to covellite, and the arrangement of the covellite flakes shows the original cleavage of galena. In this way, pseudomorphs of covellite on galena are originated. The formation of covellite in galena myrmekites with copper sulfides generates textures that simulate covellite myrmekites in galena.
Substitutions 5: pyrite is substituted for covellite, but these are rare cases.
Substitutions 6: the substitution of covellite by other minerals is rare, disregarding oxidation processes. Covellite may be replaced by chalcocite, bornite, digenite and chalcopyrite. In some cases, covellite is covered by chalcocite and acanthite during cementation processes.