FLUORITE
Fluorite – CaF2 – is a very common halide, a strategic ore, essential in steel making and metallurgy as a flux, also in the chemical industry (HF manufacturing) and for many other applications. The main source of “metallurgical grade fluorite” are sulphide ores of lead and zinc with fluorite gangue. In addition, due to the great diversity of colors and the combined cubic forms that it can display, fluorite has huge acceptance in the collectible minerals market, achieving very high prices.
Forms crystals up to 2 meters. A number of impurities can occur, such as Al, Fe, Mg, Cl and Rare Earths (Y, Ce, Eu, Sm, etc.). It sometimes fluoresces in green, yellow, violet, and other colors under UV light; it can be phosphorescent, triboluminescent and thermoluminescent. Epitaxies can occur with siderite (with [0001] of siderite parallel to [111] of fluorite), with pyrite (parallel axes of pyrite and fluorite) and with quartz. There are 10 varieties (with Y, with Sr, with Ce, etc.).
1. Characteristics
Crystal system: Cubic hexaoctaedrical.
Color: Anything from colorless to black. Often zoned.
Habit: Cubes, octahedra, combined forms, nodular, botrioidal, massive, granular, rarely columnar or fibrous.
Cleavage: {111} perfect (octahedral).
Tenacity: Brittle.
Twinning: Common in {111}, of interpenetration.
Fracture: Subconchoidal, irregular.
Mohs Hardness: 4
Parting: On {011}, of low quality.
Streak: White.
Lustre: Vitreous.
Diaphaneity: Transparent.
Density (g/cm³): 3.12 – 3.18. 3.56 if with REE.
2. Geology and Deposits
Fluorite is a common accessory mineral in granitic rocks in general and associated rocks such as greisens and pegmatites. Can occur filling fractures in these rocks. It also occurs in carbonatites, syenites, nepheline syenites, alkaline intrusions and ultrapotassic rocks. It can occur in marbles, quartzites and other metamorphic rocks.
Economic volumes occur in mineralized hydrothermal veins, from high to low temperature. It is found in cavities in sedimentary rocks and as cement in sandstones, as well as in hot spring deposits and in biogenic dolomites.
The occurrence of fluorite in granitic rocks is a problem, because the groundwater that circulates through the fractures of the granitic massif becomes enriched with fluorine. If these fractured aquifers are exploited, it is necessary to control the fluoride content to avoid health problems (fluorosis), especially in children and the elderly.
3. Mineral Associations
Fluorite occurs with quartz, feldspar (potassic and calc-sodic), mica (biotite, muscovite), carbonates (calcite, dolomite, rhodochrosite), sulfates (barite, celestine), sulfides (galena, sphalerite, pyrite, chalcopyrite), scheelite, apatite, cassiterite (in greisens), topaz, tourmaline and wolframite (in pegmatites).
4. Transmitted Light Microscopy
Refraction indices: n = 1,433 – 1,448
PLANE POLARIZED LIGHT – PPL
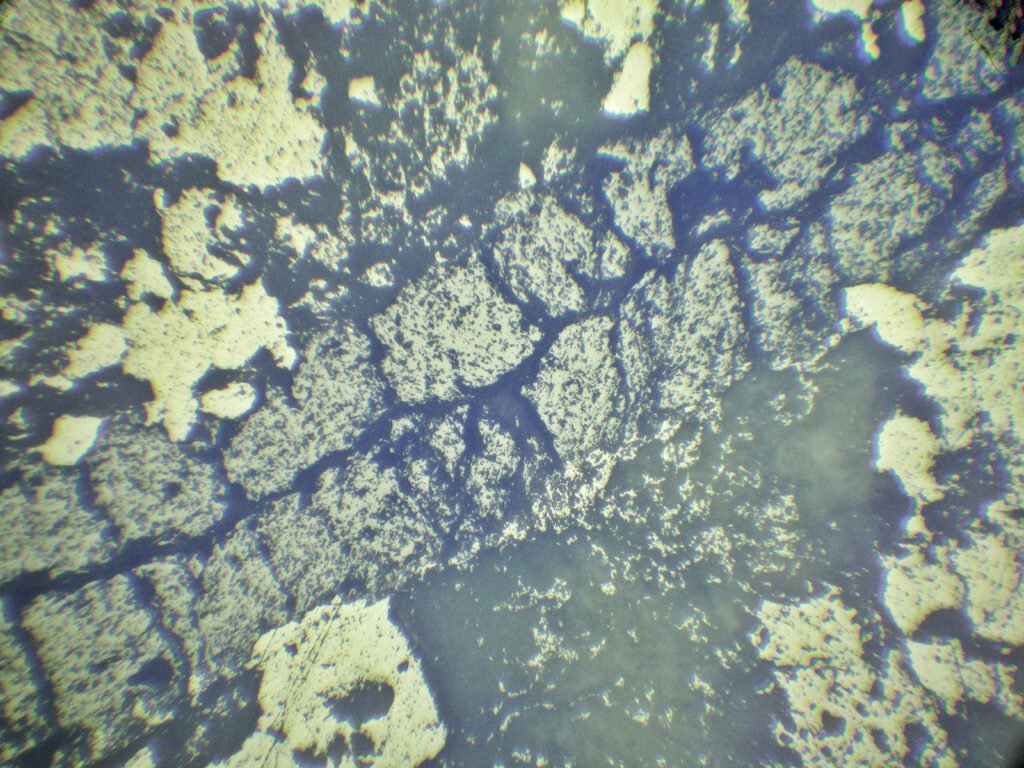
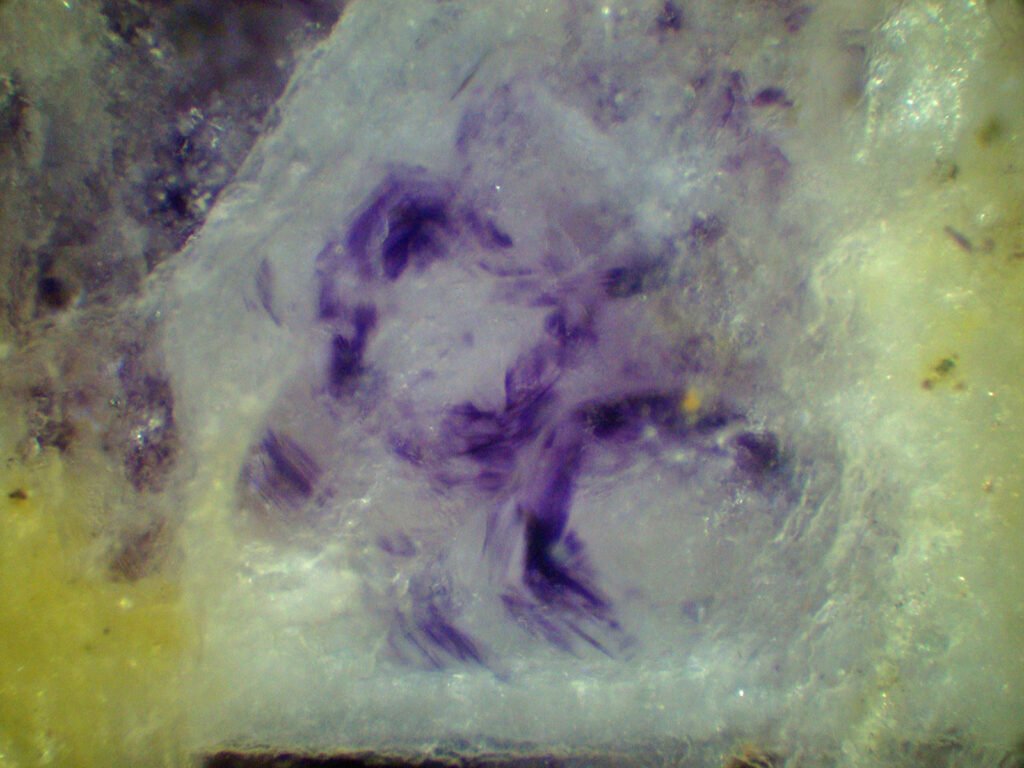
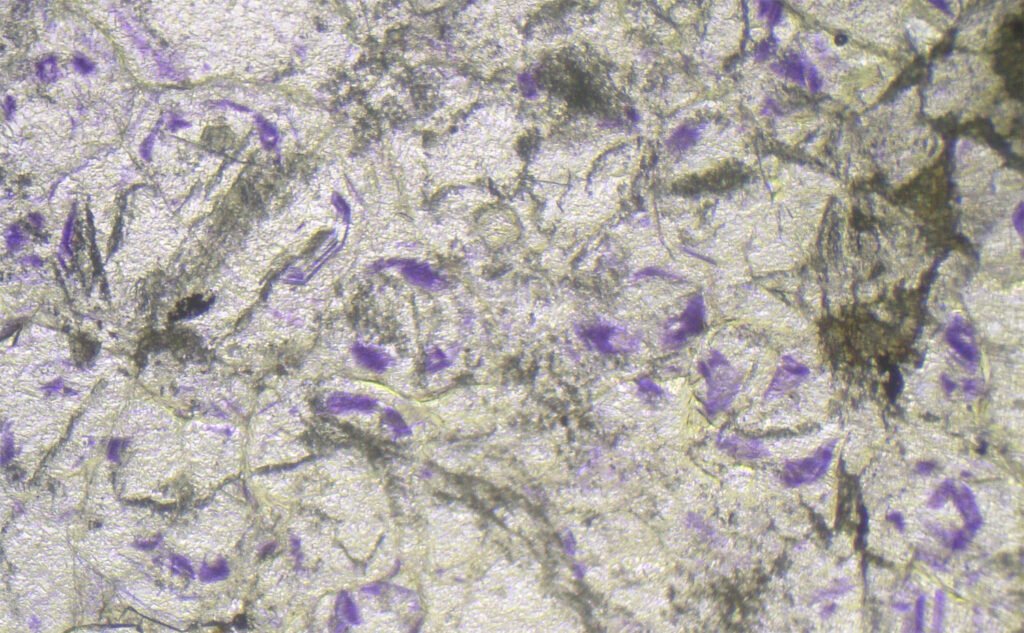
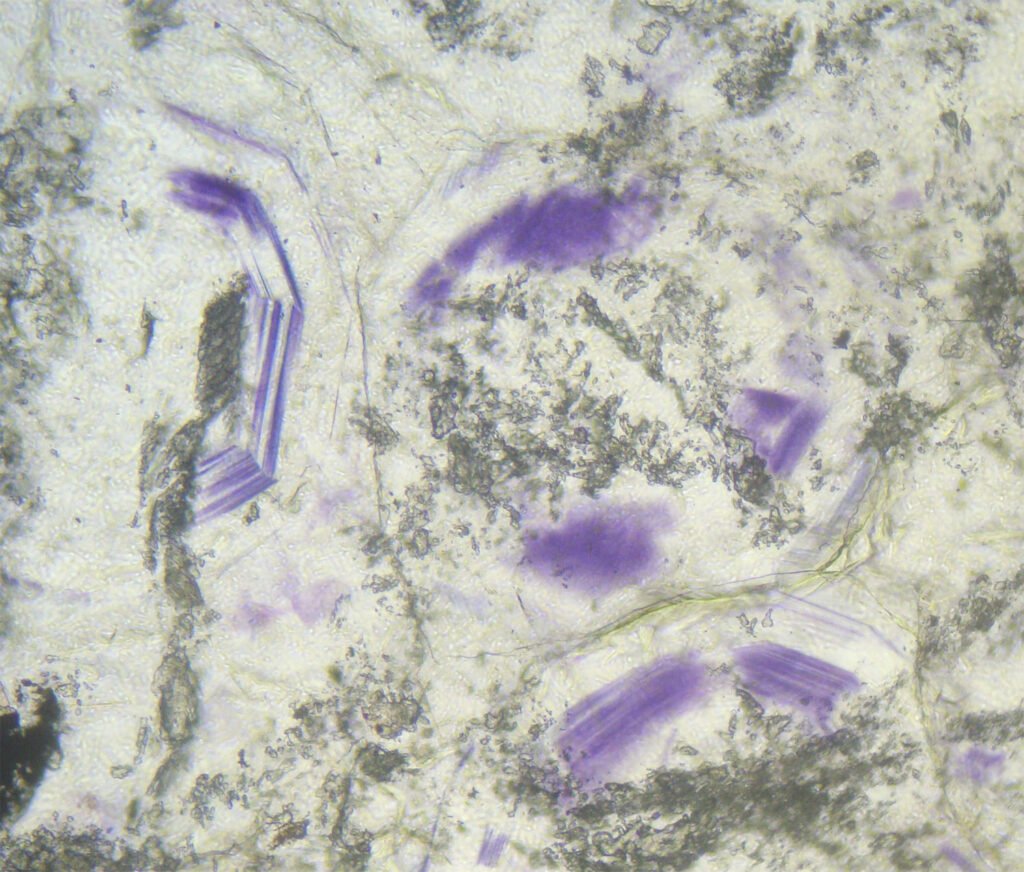
Color / Pleochroism: Colorless (colored in thick sections), rarely pale violet or pale green. It may be zoned (purple edges, for example) or with irregular color patches.
In purple fluorites, colored halos around radioactive inclusions are typical.
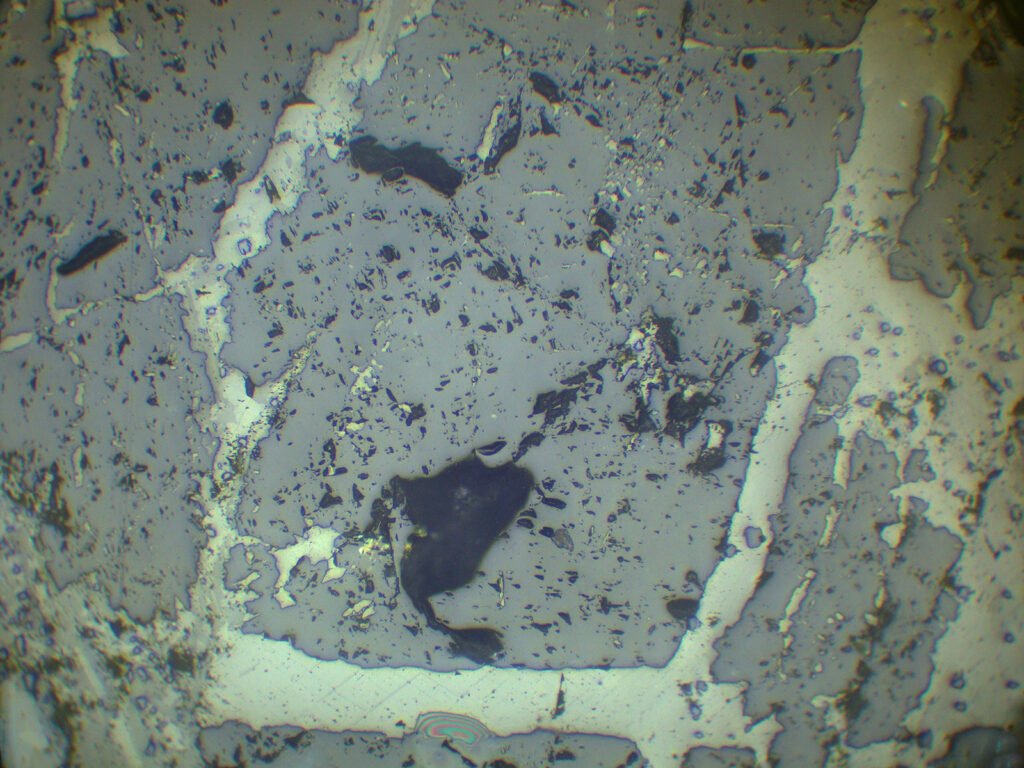
Relief: Moderate (or high negative, according to other sources).
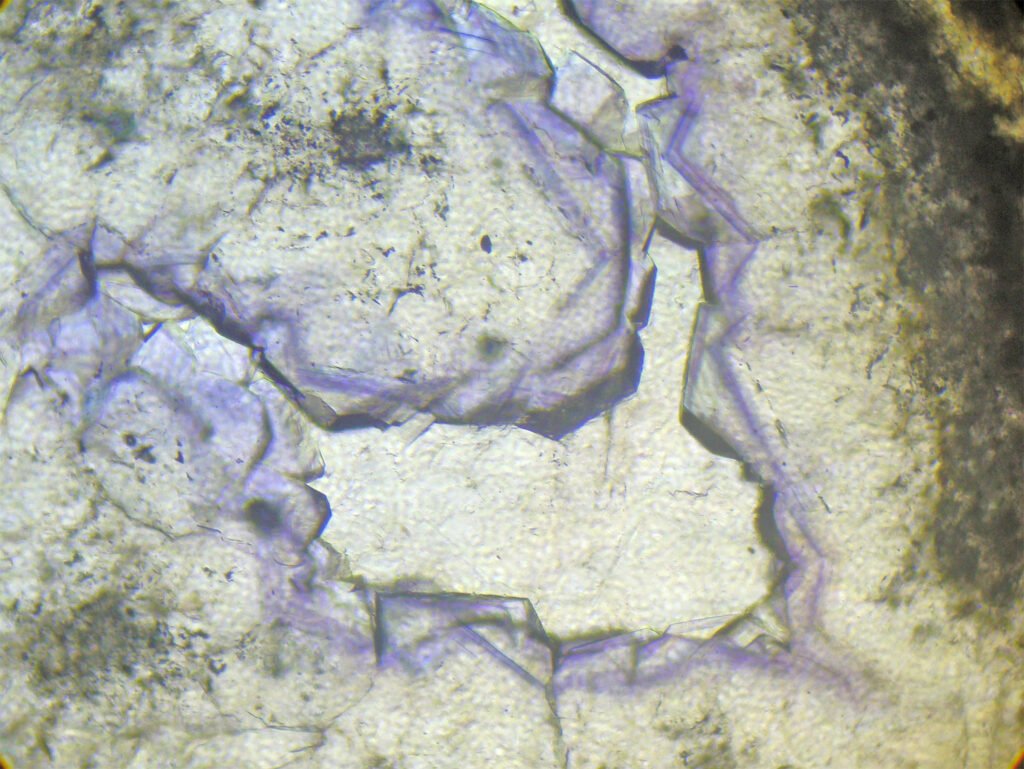
Cleavage: {111} perfect (octahedral – form triangles).
Cleavages intersect at angles of 70° and 110°, or as 3 lines intersecting at angles of 60° and 120°. If microcrystalline does not show cleavage!
Habits: Usually anhedral, rarely cubic forms. Granular, 6-sided sections, as interstitial material between previously formed minerals.
CROSSED POLARIZED LIGHT – XPL
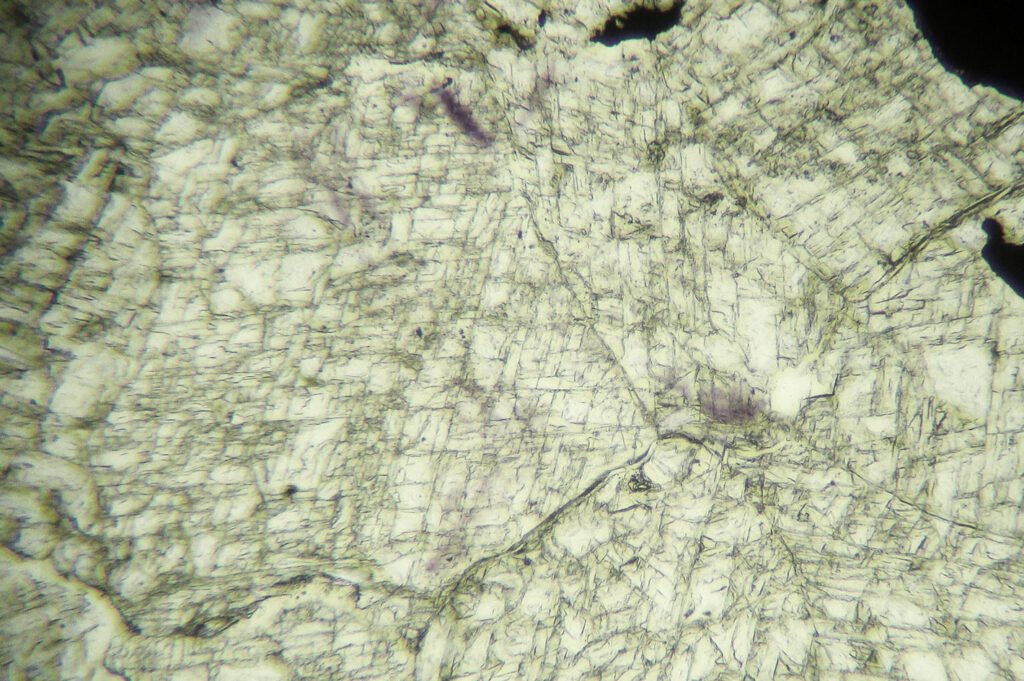
Birefringence and Interference Colors: Isotropic. May have very weak anomalous birefringence, especially in cleaved, cut, or pressed crystals. This birefringence is distributed in lamellae parallel to [001].
Extinction: Isotropic.
Elongation sign: Isotropic.
Twins: Common, on (111) (octahedral), interpenetration twins, difficult to see under the microscope.
Zoning: Often zoned, in portions (dots, bands, zones).
CONVERGENT LIGHT
Character: Isotropic.
2V angle: Isotropic.
Alterations: does not alters easily, it can even be debrital (occurs in sands).
May be confused with: other common isotropic minerals. Purple spots or bands (zoning) due to uranium and thorium traces are very characteristic. Isotropy, moderate relief, color zonation and perfect cleavage are diagnostic.
Sodalite has much lower relief.


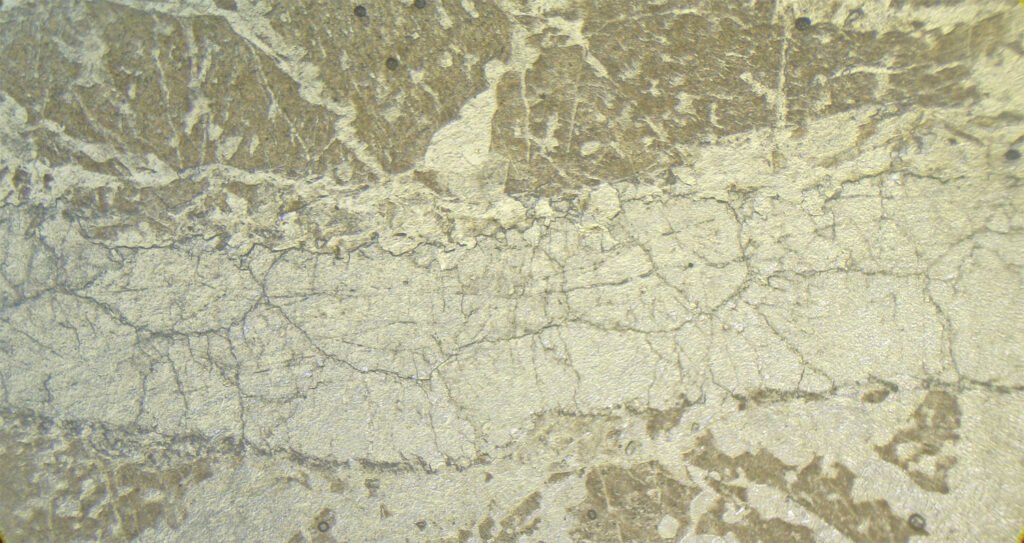





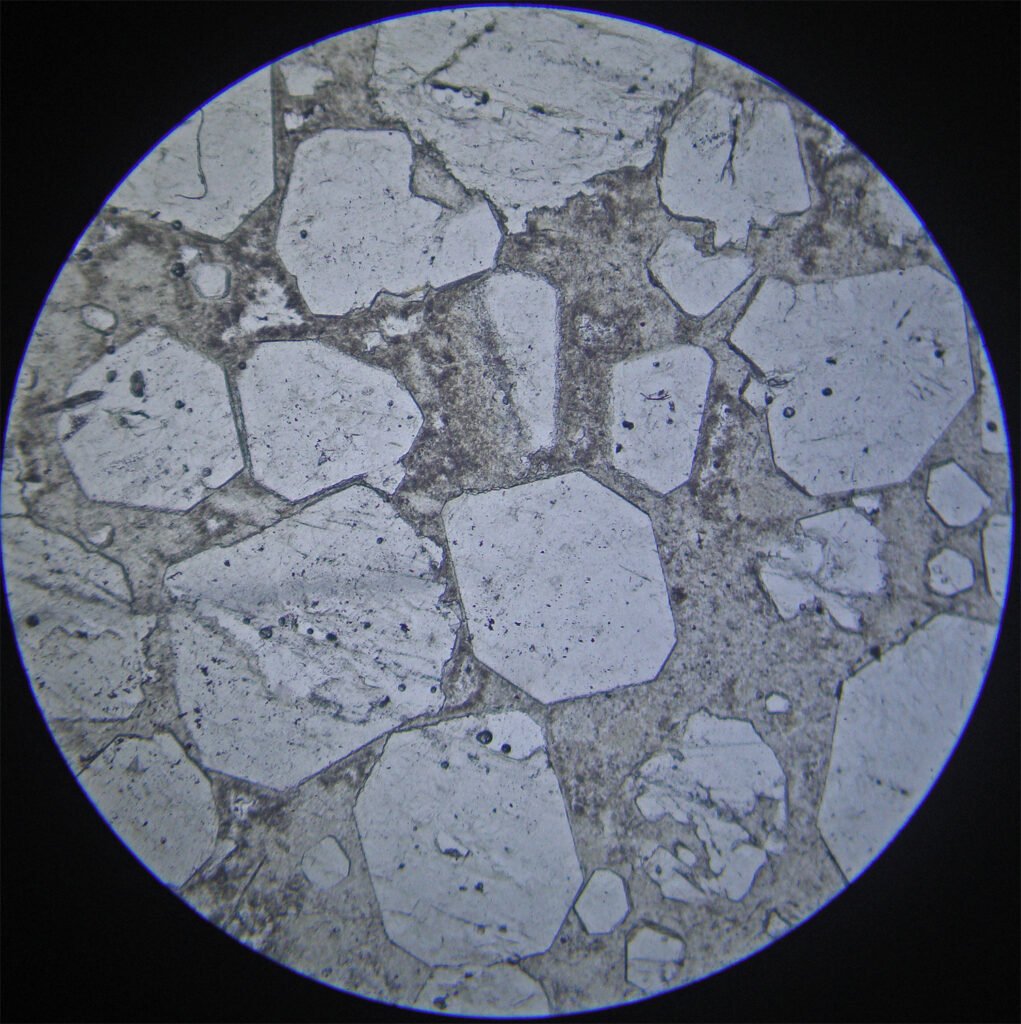
5. Reflected Light Microscopy
Reflected light microscopy is not the recommended analytical method for the identification of fluorite. However, it is important to make a polished thin section or a polished section to identify the opaque minerals that occur associated with fluorite.
Sample preparation: Fluorite polishing is simple and is of very good quality. Fluorite is slightly harder than calcite; it has approximately the same polishing hardness as chalcopyrite.
PLANE POLARIZED LIGHT – PPL
Reflection color: Dark gray, darker than common rock-forming minerals like quartz and feldspar.
Pleochroism: No.
Reflectivity: Very low (4%).
Bireflectance: No.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: Isotropic.
Internal reflections: Generalized, in the macro color of the mineral, they can be colorless, green, yellow, blue, violet, pink and others.
May be confused with:
Gypsum is very similar, but it is much softer and will always have many polishing scratches.
Zeolites may be similar, but their habits are different.
General Characteristics:
Cleavage: perfect octahedral cleavage (in 3 directions) gives rise to triangular polishing pits if the fluorite grain is coarse and the polish is of poor quality. As also happens with galena, microcrystalline aggregates do not show polishing pits.
Twins were not observed.
Zonation: color zonation is very common.