GALENA
Galena – PbS – is a very common sulphide, the main ore of Pb and often an important ore of Ag. Pb-Zn ores generally contain economic grades of Cu, Ag, Sb and Bi.
Galena always contains varying, usually very low, levels of Ag, Cu, Fe, Tl, Mn, Zn, Cd, Sb, As and Bi. Many of these “impurities” can actually come from tiny inclusions of other minerals. There are two solid solution series, one with clausthalite (PbSe) and the other with altaite (PbTe). There are a dozen varieties, based on expressive levels of certain elements or on specific habits. Galena is a semiconductor, so galena crystals were used as diodes in “galena radios”.
Galena crystals are common. They usually form cubes (up to one meter edge), they can form octahedrons or complex combined forms. The typical cubic cleavage of galena changes if other elements are present: with Bi the cleavage is octahedral, with Ag the cleavage becomes scaly. Newly cleaved crystals exhibit faces with a strong metallic luster, which over time can tarnish, lose their luster and become dull. There are contact and interpenetration twins by {111} in addition to lamellar ones by {114}.
Contains lead, which is toxic: always wash your hands after handling. Avoid inhaling the dust when you are breaking down the mineral. Do not lick; much less ingest. Lead poisoning, both chronic and acute, was and continues to be common, due to the use of Pb in creams, bottles, water pipes, etc.
1. Characteristics
Crystal system: Cubic, hexaoctahedral.
Color: Lead gray to white, from light to dark, with a shade of blue. Tarnishes in the air.
Habit: Crystals, massive, coarse to fine granular, tabular, skeletal, fibrous, feathery.
Cleavage: {001} perfect.
Tenacity: Brittle.
Twinning: See above.
Fracture: Subconchoidal, straight
Mohs Hardness: 2.5
Parting: On {111}.
Streak: Dark gray to black.
Lustre: Metallic, dull.
Diaphaneity: Opaque.
Density (g/cm³): 7.2 – 7.6
2. Geology and Deposits
Galena forms in a number of environments. It occurs in hydrothermal veins with sulfides formed over a wide range of temperatures. It is one of the main ore minerals in Pb-Zn-Cu deposits of the VMS (Volcanogenic Massive Sulphides), MVT (Mississippi Valley Type) and SEDEX (Sedimentary Exhalative) types. It also occurs in contact metamorphism deposits such as skarns.
It occurs disseminated as small and sparse grains in igneous rocks. In sedimentary rocks it can replace carbonates or occurs as cement in clastic rocks such as sandstones. It is rare in placers (due to its low hardness) and in pegmatites.
3. Mineral Associations
Occurs associated with classic gangue minerals, such as quartz, carbonates (calcite, dolomite, siderite), fluorite, barite and others.
In hydrothermal veins, a very common association is with pyrite, marcasite, sphalerite, chalcopyrite, arsenopyrite, magnetite and other common sulfides such as tetrahedrite-tenanntite and bournonite. In silver mineralization, galena is very common, associated with a wide variety of silver minerals.
In the oxidation and alteration zones, galena remains are associated with cerussite, anglesite, pyromorphite and witherite. Generally speaking, galena is associated with at least 50 different minerals.
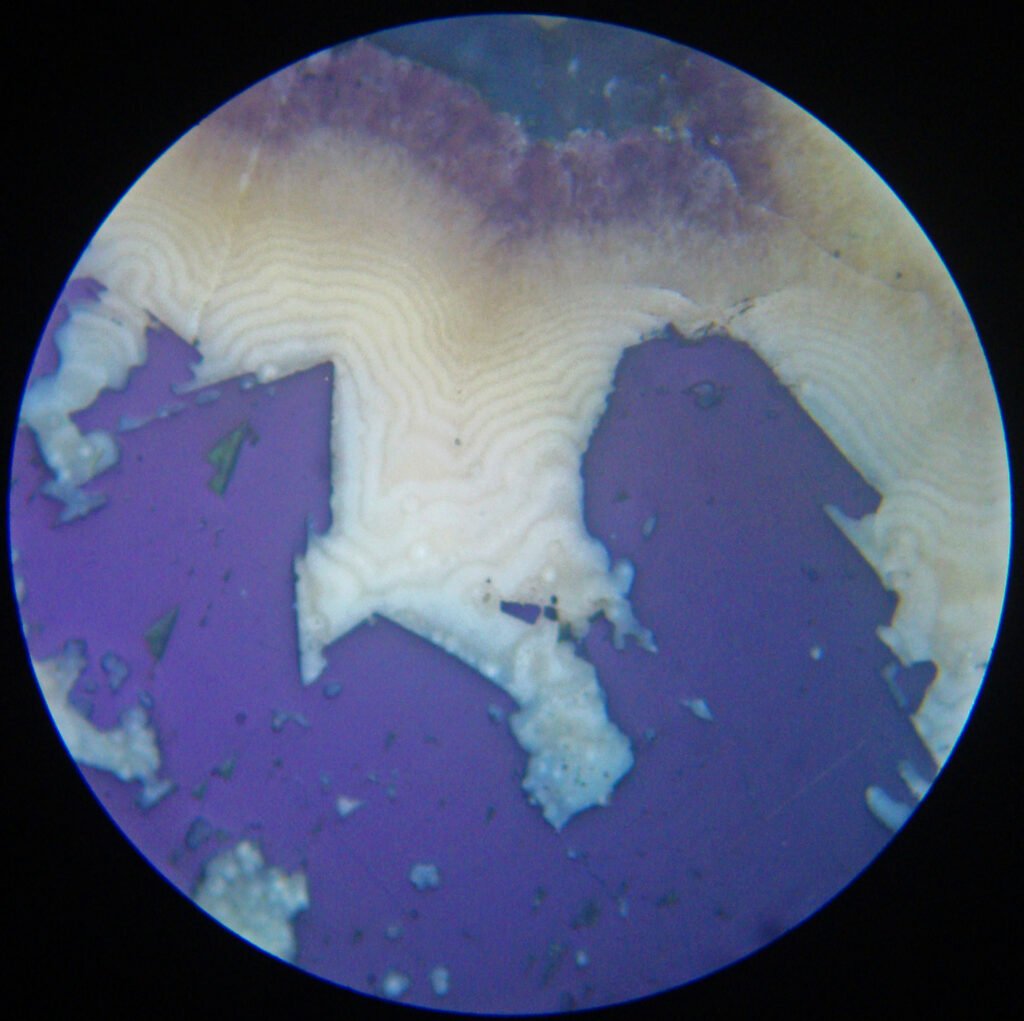
4. Transmitted Light Microscopy
Not applicable, as galena is completely opaque.
In some cases, however, when the galena shows large crystals, the same triangular polishing pits, well-known from the polished sections in Reflected Light, may appear on the thin section.
The existence of triangular and square holes in an “opaque”, therefore, strongly suggests the existence of galena and it is possible to confirm this indication using the Oblique Light technique.


5. Reflected Light Microscopy
Sample preparation: the polishing of galena is hampered by the low hardness of the mineral. The polishing hardness of galena is greater than that of acanthite, proustite, pyrargirite and covellite. It is equivalent to that of bournonite and very similar to the hardness of chalcocite. It is much lower than those of chalcopyrite, tetrahedrite-tennantite and the vast majority of silicates and ore minerals.
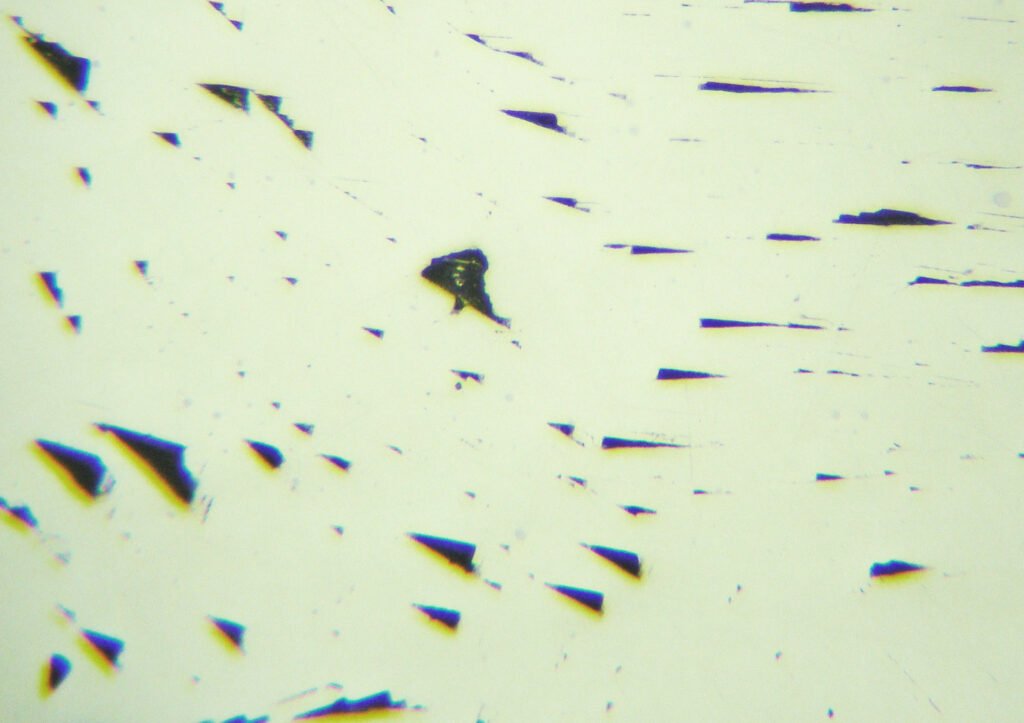
In this way, polishing scratches will be unavoidable and care must be taken to prevent the galena from having a strong negative relief in the polished section (use a “hard” cloth and not a “soft” cloth; avoid the use of coarse abrasives in the initial grinding ). When galena occurs in large crystals, fragments are loosened during polishing (producing triangular polishing pits or not) that scratch the surface of the entire polished section or thin section.
PLANE POLARIZED LIGHT – PPL
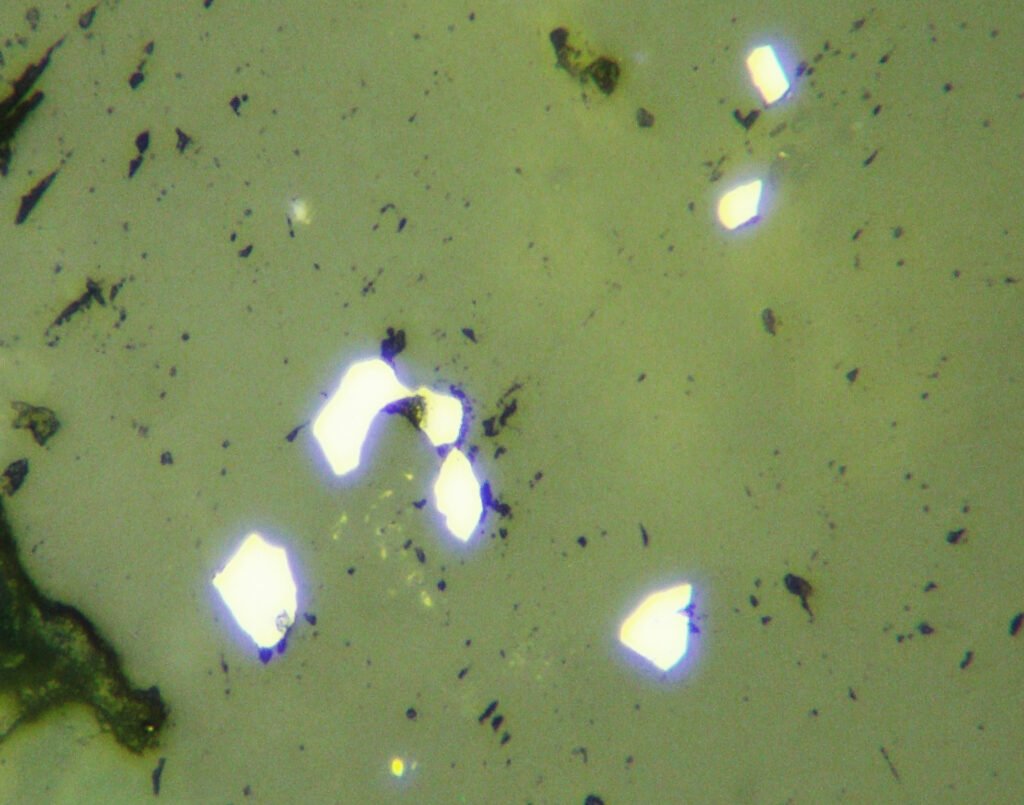
Reflection color: White to gray-white (this is the standard white of Reflected Light Microscopy, used in Murdoch’s Tables). It is the whitest ore mineral of many ores. As galena is widely distributed, its color is very useful as a color reference in complex ores.
Very small amounts of tellurium change the color to red and purple.
Extremely fine-grained galena shows a much darker color than large-grained galena: the color is between bronze and gray.
If the ore does not contain galena, it is useful to use another section with galena as a reference to “calibrate” the eye before starting observations.
Compared to the color of sphalerite, the color of galena is white.
Compared to the colors of tennantite, bismuthinite and bournonite, the color of galena is pink.
Compared with the color of stibnite, the color of galena is much lighter.
Pleochroism: No.
Reflectivity: 41.56%
Bireflectance: No.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: Isotropic. The ever-present polishing scratches can simulate anisotropy.
Non-stoichiometric or deformed crystals are slightly anisotropic between medium gray and gray-black.
Internal reflections: No.
May be confused with: if galena occurs in large grains, showing the triangular polishing pits, it can only be confused with a few minerals, usually rare, if the polished section is of good quality. Triangular polishing pits in this quality and quantity only appear in clausthalite, altaite and sylvanite (slightly yellow).
However, when galena occurs in very small grains or aggregates of very small grains, it can be confused with many other minerals (native silver, sulfides, and sulfosalts), which have similar color and reflectivity. Many of these, however, are anisotropic and not isotropic like galena. The verification of this anisotropy, on the other hand, can be impossible or very difficult when the grains are very small.
General Characteristics:
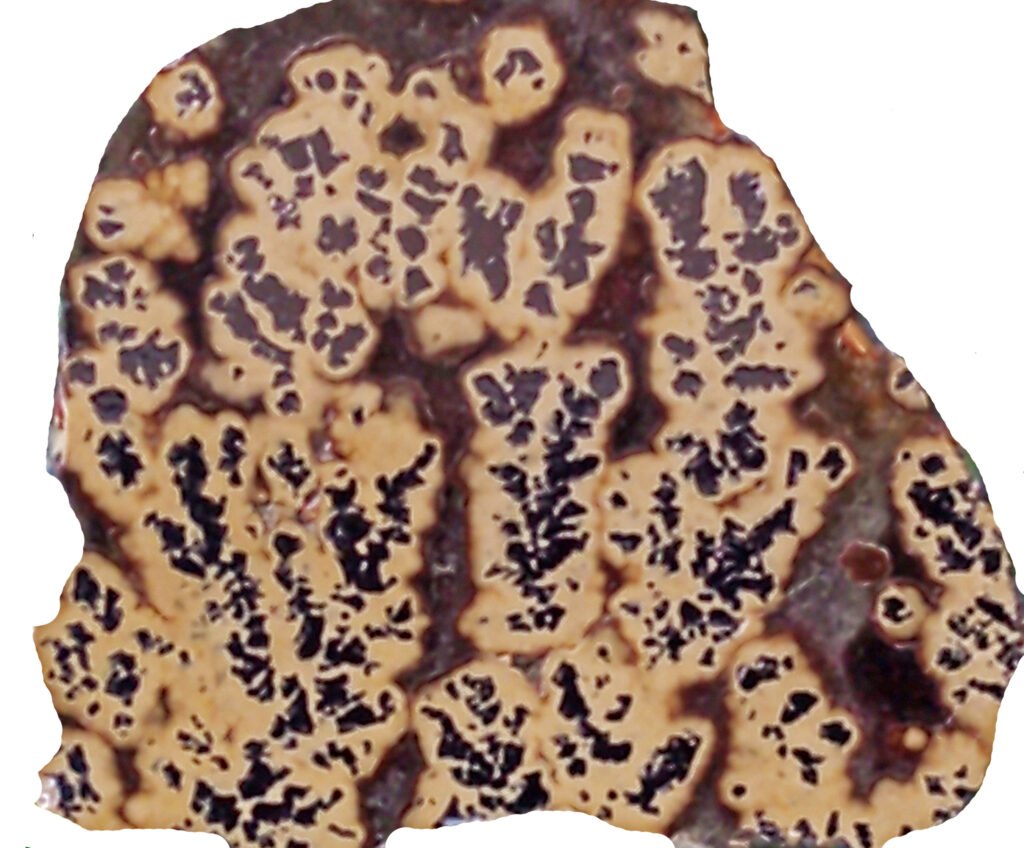
Grain shape: Galena easily forms large, idiomorphic crystals, especially when growing freely in cavities, for example. Frequent, however, are very fine-grained masses or very small isolated and anhedral grains, both of which are difficult to identify. Generally, galena forms masses without a defined shape. It may be skeletal (dendritic) and encased in finely banded sphalerite. “Knitted Galenas” are skeletal (acicular) crystals that grew along the edges of the cube, often with continuity for up to 5 cm. The boundaries of large, often discernible grains are formed by the faces of the octahedrons. Tectonic processes subsequent to the formation of galena can destroy the original idiomorphy, generating several different textures. Recrystallization, on the other hand, has the opposite effect, with the formation of new crystals that are larger, less deformed and fractured and more idiomorphic. This ease of galena to alter its shapes suggests care in interpreting ore textures and explains the contrast between galena textures and the textures of other associated less plastic ore minerals.
Polishing scratches due to low hardness are always present; it is almost impossible to produce a galena-polished section without polishing grooves, which become more visible in CPL.
Low relief (negative) occurs in the presence of harder neighboring minerals such as most sulfides and silicates that associate in the paragenesis. As a result, in the probable presence of galena, greater care is recommended in the production of the polished section, precisely to avoid this low relief, which hinders observations.
Triangular polishing pits are formed by the detachment of cleavage fragments and are important for recognizing galena. However, this only occurs when the galena presents itself in larger crystals. Very fine-grained galena masses will not show the polishing pits, generally show a good quality polish, and can be confused with dozens of white, highly reflective and isotropic or low anisotropic minerals.
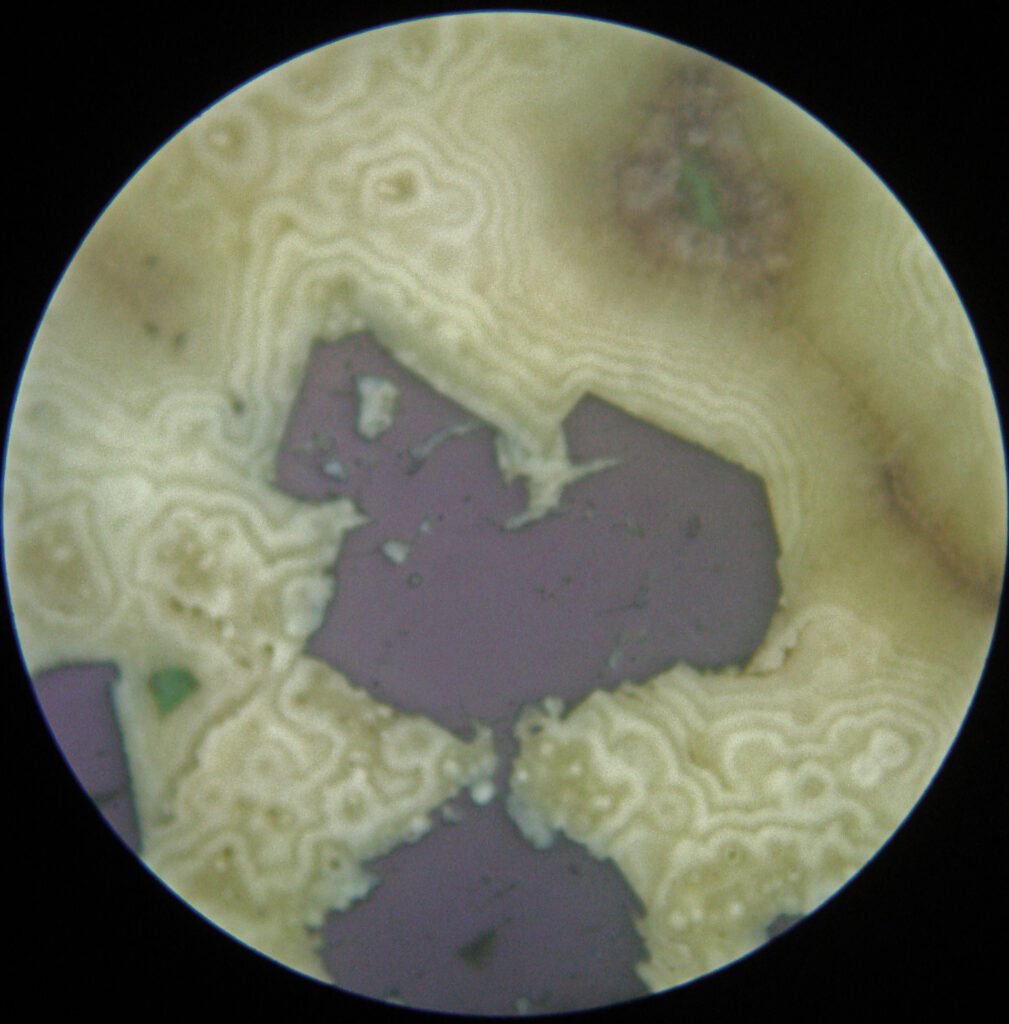
Perfect cubic cleavage is visible if galena forms large crystals. Cleavage is often highlighted by fracturing that followed cleavage plans, with the introduction of secondary minerals. Anomalous cleavage parallel to {111} is due to Bi contents.
Inclusions are frequent, they can be of boulangerite, bournonite, pyrargyrite and tetrahedrite-tennantite. The most common are bournonite (with the typical twins) and tennantite-tetrahedrite (isotropic).
Deformation lamellae (deformed grains) can occur and are evidenced by sinuous alignments of triangular polishing pits, historically called “Bleischweif” (“lead tail” in German). They are thin, tabular and parallel crystals.
Pseudomorphoses on galena can be of several secondary minerals such as chalcocite, anglesite, cerussite, covellite and pyromorphite. In turn, galena forms pseudomorphoses on cylindrite, franckeite and teallite when in almost graphic intergrowths with cassiterite and boulangerite.
Twins occur according to various laws in isolated crystals, but they do not occur in massive galenas.
Zonation, sometimes excellent, is often visible through chemical attack or by incipient alteration. Zonation can form by the zonal distribution of submicroscopic pores or by growth interruptions, during which submicroscopic bodies of other minerals, gas bubbles, and the like are deposited on the surfaces of the crystals at each stage.
Deformations are common, both in isolated grains and in aggregates, due to the plasticity of galena. They are simple displacements associated with translation according to several directions; usually several of these occur together. Great density of these “twin lamellae by pressure” occurs in the “Bleischweif” (“lead tail”).
Cataclasis is not very frequent, as the deformation is easily processed without fracturing, generating twins and curved alignments of the polishing pits.
Intergranular contacts, in unaltered occurrences, are very interfingered on a fine and delicate scale. By recrystallization this entanglement is lost.
Recrystallization of galena, by high temperature events, produces a granoblastic aggregate of uniformly sized grains. If the event is delayed, continued growth causes the granoblastic texture to disappear again. In these cases, the only evidence of the process are the pieces generated by the cataclase of the less plastic associated minerals, which are still present and unchanged.
Oriented intergrowths occur with pyrrhotite, millerite, chalcopyrite, tetrahedrite, native bismuth, native antimony, chalcocite, acanthite, tetradymite, altaite, polybasite, covellite, pyrargyrite, bournonite, jamesonite, galenobismutite, pyrite, arsenopyrite, geochronite, ullmannite, quartz, other silicates and “schapbachite” (eg, matildita + galena). These very fine textured intergrowths create complicated problems in the ore beneficiation process.
Inclusions are very common, as galena is usually of late formation in the paragenesis. Inclusions can be (i) from older minerals, (ii) substances introduced later, especially at fractures, and (iii) substances that are likely to be unmixings (e.g., tetrahedrite-tennantite, pyrargyrite and others).
Silver contents have an opposite behavior to that of lead: in the processes of galena alteration, Pb is quite immobile and forms secondary lead minerals. Silver, on the other hand, is easily removed in the form of carbonate or Ag sulfate and deposited further down in the cementation zone of the deposit, forming native silver and acanthite in fractures and other types of discontinuities. Very rarely does silver remain in the original position (on the top of the deposit) in the form of chlorargyrite or as native silver inclusions in cerussite. There is an extensive literature on the origin of silver contents in galena, but it is usually due to very fine inclusions of many different silver-bearing minerals, such as tetrahedrite-tennantite, pyrargyrite, proustite, native silver, acanthite, dyscrasite, stromeyerite, pearceita, stephanite and others, with silver-bearing minerals varying from deposit to deposit.
Redeposition of galena is rare, but Pb can be transported from the top of the deposit down to the cementation zone, where it forms (i) galena with very well-developed rhythmic rearrangement structures associated with native sulfur or (ii) galena with skeletal growths and porous nuclei alongside normal older galenas.
Myrmekites are frequent with galena forming myrmekites with chalcocite, stromeyerite, tetrahedrite-tennantite and chalcopyrite.
Gel structures are rarer due to the great ease of crystallization of galena. Even so, they are relatively frequent, known from several locations.
Substitutions are very common in galena and must be discussed by its environment: of alteration or of formation of galena. (i) Alteration substitutions occur in the oxidation zones (“gossan”) and in the cementation zone of sulphide deposits including galena. (ii) Formation replacements occur in those geological environments where galena is introduced by hydrothermal processes.
Substitutions 1: as galena alteres very easily under common atmospheric conditions, substitution occurs by alteration minerals, mainly cerussite, secondarily by anglesite, also pyromorphite, wulfenite and many others; they are usually several together side by side. These new minerals encapsulate the galena and protect it from further alteration. They also penetrate the galena through cleavage planes and sometimes along zone boundaries. Recognition of these under the microscope is quite complicated, as their optical characteristics are very similar.
Substitutions 2: galena may be replaced by covellite and chalcocite, both in the oxidation zone and mainly in the cementation zone. The process can be complete, generating pseudomorphoses of covellite on galena. This covellite can be arranged randomly or the lamellae are oriented parallel to (100) of the galena.
Substitutions 3: Galena can be replaced by a number of other minerals such as pyrite, pyrrhotite, tetredrite-tennantite, enargite, sphalerite, chalcopyrite and even magnetite. More rarely, it is replaced by native silver. Several generations of galena can occur in the deposit, so care must be taken in the interpretation of textures.