GOLD
Gold – Au – is a native element that constitutes ore of gold and other elements. Aesthetic pieces fetch prices much higher than the standard price of gold in the collector minerals market.
Gold always forms mixing crystals. It forms a series with silver and usually contains Ag, Cu and Fe, more rarely I, Ti, Ni, Pb, Sb, Hg, V, Bi, Mn, As, Sn, Zn, Pd, Pt and Cd. There are a dozen varieties, always based on the content of other elements. “Electrum” is gold with 30-45% silver; “küstelite” is gold with 80% Ag; “porpezite” and “palladian gold” is gold containing Pd; “rhodite” is a variety with Rh, etc. In some deposits all the gold occurs under the microscope. Some ores, even very rich, show practically no visible gold, even in the absence of tellurides and gold selenates. In such cases the ore may contain up to 1 kg of gold per ton without any visible gold; gold is present in the form of grains with diameters of 0.01 microns.
Gold generally occurs in anhedral grains. Crystals are relatively rare and are often distorted. The largest crystals reached 5 cm, can form twins and parallel associations. Gold is diamagnetic (repelled by magnetic fields).
The “Acid Test” is based on the fact that gold does not dissolve in nitric acid, unlike silver and base metals. Gold is also dissolved in aqua regia (nitric acid + hydrochloric acid), dissolves in alkaline cyanide solutions, and dissolves in mercury, forming amalgam.
1. Characteristics
Crystal system: Cubic hexaoctahedral.
Color: Pure: golden yellow with a red tint. Impure: silver-white to copper-red. Very fine powder: brown.
Habit: Crystals (distorted octahedrons and cubes) rare; dendritic, granular, nuggets, vanes, etc.
Cleavage: No.
Tenacity: Very ductile and malleable.
Twinning: {111} common.
Fracture: Hackly.
Mohs Hardness: 2.5 – 3
Parting: No.
Streak: Shining yellow.
Lustre: Very strong metallic.
Diaphaneity: Opaque, very thin leaves are translucent.
Density (g/cm³): 15.6 – 19.3 When pure: 19.3
2. Geology and Deposits
Primary gold deposits are of two types. Hydrothermal quartz veins and related deposits contain widespread gold grains, dendrites and wire gold aggregates and are associated with igneous and metamorphic rocks. In volcano-exhalative deposits, on the other hand, gold occurs associated with sulfides.
Secondary gold deposits are elluvial and alluvial deposits (gravel, sand) where gold has concentrated due to its high density.
3. Mineral Associations
Gold occurs associated with quartz, tourmaline and many dozens of sulfides (see below). Also with the typical minerals of the Au-Ag-Te-Hg association such as sylvanite, krennerite, calaverite, altaite and others.
4. Transmitted Light Microscopy
The literature reports that under Transmitted Light native gold is blue and green.
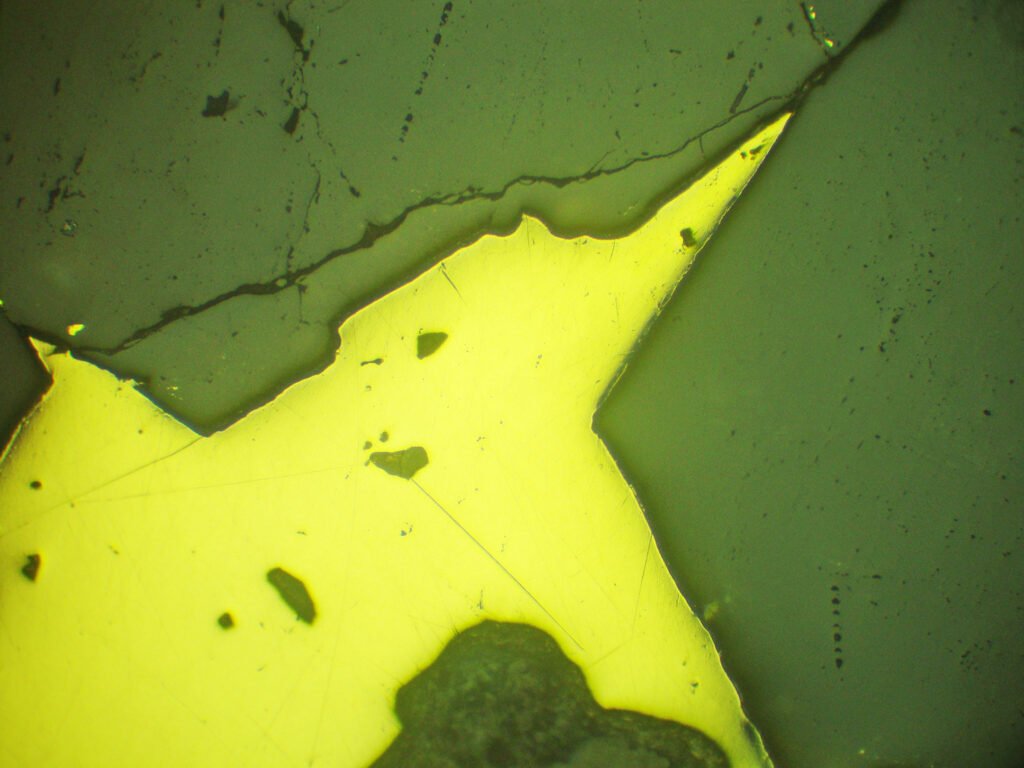
5. Reflected Light Microscopy
Sample preparation: Pure native gold acquires a good polish with ease. Polishing scratches are difficult to avoid, practically impossible, somewhat reminiscent of native silver. Applying high pressures during grinding and pre-polishing should be avoided as much as possible, as abrasive particles penetrate the gold and from there prevent a good polishing, in addition to providing erroneous results in analytical equipment (electron microprobe). The polishing hardness is greater than the hardness of galena and similar to that of chalcopyrite.
PLANE POLARIZED LIGHT – PPL
Reflection color: The reflection color varies greatly depending on the contents of other elements.
Gold with low silver contents is bright yellow gold.
Gold with high levels of silver is pale yellow to silvery white.
Copper-rich gold is pink to copper red.
Palladium-rich gold is almost creamy white.
Compared with the color of electrum, the color of gold is more yellow.
Compared with the color of chalcopyrite the color of gold is paler and lighter yellow.
Compared to the colors of silver, bismuth and platinum, the color of gold is much more yellow.
Pleochroism: No.
Reflectivity: Very high if pure (72.26%). Electrum: 92.3% (Au,Pd): 66.6%
Bireflectance: No.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: Isotropic. However, it is never completely dark, but shows some strange greenish hues, something like dark citrus green, especially along the polishing scratches.
In XPL, all the polishing scratches appear below the surface film (Beilby layer), simulating anisotropy.
Internal reflections: No.
May be confused with: the high reflective power, the low hardness (many polishing grooves!) and the extremely vivid color exclude mistakes. Very small grains of pyrite or chalcopyrite in gangue minerals can look like gold, especially if viewed in the air (without oil immersion techniques). Silver blurry grains can be very similar. Very small grains of native copper can also be very similar to native gold.
General Characteristics:
Grain shape: It is usually granular isometric with very variable contacts, from smooth to toothed. Reniform-botryoidal clusters occur, sometimes with rhythmic banding. In these bands, the isolated grains are very small or submicroscopic. Large grains can reach sizes up to one cm. Fine granular levels may recrystallize, erasing the original features. Common habits are skeletal, thin acicular, colloidal shapes, crusts around pyrite, chalcopyrite and galena and many others.
When in the midst of other minerals, the individual grains are usually anhedral; well-formed crystals (pentagons, dodecahedral, pseudohexagonal) are found only occasionally in galena, stibnite and evidently in pores and druses. Very common are thin sheets of gold that occur as ligands in cataclastic pyrites and arsenopyrites, also occasionally in tetrahedrite-tennantite and sphalerite. Similarly, gold occurs at the edges or fractures of quartz grains. In quartz-gold veins the gold grains often have sizes on the limit of microscopic visualization, in small grains and drops in quartz, pyrite or arsenopyrite.
Varieties of gold do not just occur as separate grains, but are often interwoven with one another in complex patterns, such as grains of pure gold encased in copper-rich gold.
Solid solutions of gold can occur with pyrite, arsenopyrite and sphalerite.
Cleavage is not present.
Lamellar twins arranged parallel to (111) are common; may be deformed due to tectonic stresses. Zonations are frequent in isolated crystals and are evidenced by different shades of color.
Deformations are frequent due to the strength of gold, associated with its translational capacity.
Unmixings are extremely rare, as most gold occurrences are hydrothermal. However, native gold with copper-rich skeletal lamellae arranged parallel to (100) has been observed.
Substitutions are very frequent. Gold can replace pyrite, pyrrhotite, arsenopyrite, galena, nickeline, maucherite, cubanite, millerite, gersdorffite, tellurides of Ag, Au and Hg and several other sulfides besides those mentioned above.
Inclusions of gold can occur in pyrite, marcasite, arsenopyrite, galena, sphalerite, chalcopyrite, native bismuth, bismuthinite, pyrrhotite, pentlandite, wittichenite, tetrahedrite-tennantite, freibergite, argentite, stephanite, stibnite, several different tellurides, native copper, skutterudite, bournonite, magnetite and safflorite. Isolated gold grains can occur in galena, sphalerite, or chalcopyrite, but it is possible that only one grain of these minerals has a gold inclusion, while the others nearby have no gold inclusion.
Intergrowths of gold occur with tetradymite, clausthalite, altaite and other tellurids. Myrmekitic intergrowths occur with native bismuth, thanks to the decomposition of maldonite (Au2Bi).
Secondary gold can occur in chalcocite, digenite, covellite, goethite and hematite.