
HALITE
Halite – HCl – is a halide with a restricted but worldwide distribution. It has been extremely important since the Neolithic as a salt ore and later as a base for obtaining chlorine. When combined with K and Mg salts, it also forms an ore of these elements.
May contain I, Br, Fe and O. There is an argentiferous variety (Huantajayita). May fluoresce red under shortwave ultraviolet light.
Analysis of halite under Transmitted Light is complicated by the fact that it is soluble in water (see below). Reflected Light and Oblique Light techniques do not apply.
1. Characteristics
Crystal system: Cubic hexaoctahedral.
Color: Colorless, white, can be pink, red, yellow, orange, blue or purple.
Habit: Cubic crystals, rarely octahedra, may be skeletal and cavernous (“hopper”) Rarely granular, massive, compact.
Cleavage: {001} perfect.
Tenacity: Brittle.
Twinning: On {111} on artificial crystals.
Fracture: Conchoidal.
Mohs Hardness: 2.5
Parting: No.
Streak: White.
Lustre: Vitreous.
Diaphaneity: Transparent.
Density (g/cm³): 2.168
2. Geology and Deposits
Halite is a mineral characteristic of evaporites, which are biochemical and chemical rocks formed from both salt lake water and sea water. Once deposited in layers and buried at great depths, there are conditions for the formation of salt domes, which constitute the main deposits of salt on the planet. Salt has been a strategic ore since Neolithic times.
Salt domes can rise to the surface, forming salt springs (as in a case in Germany) or even form salt “glaciers” down the mountain, when in arid climates (as two occurrences in Iran).
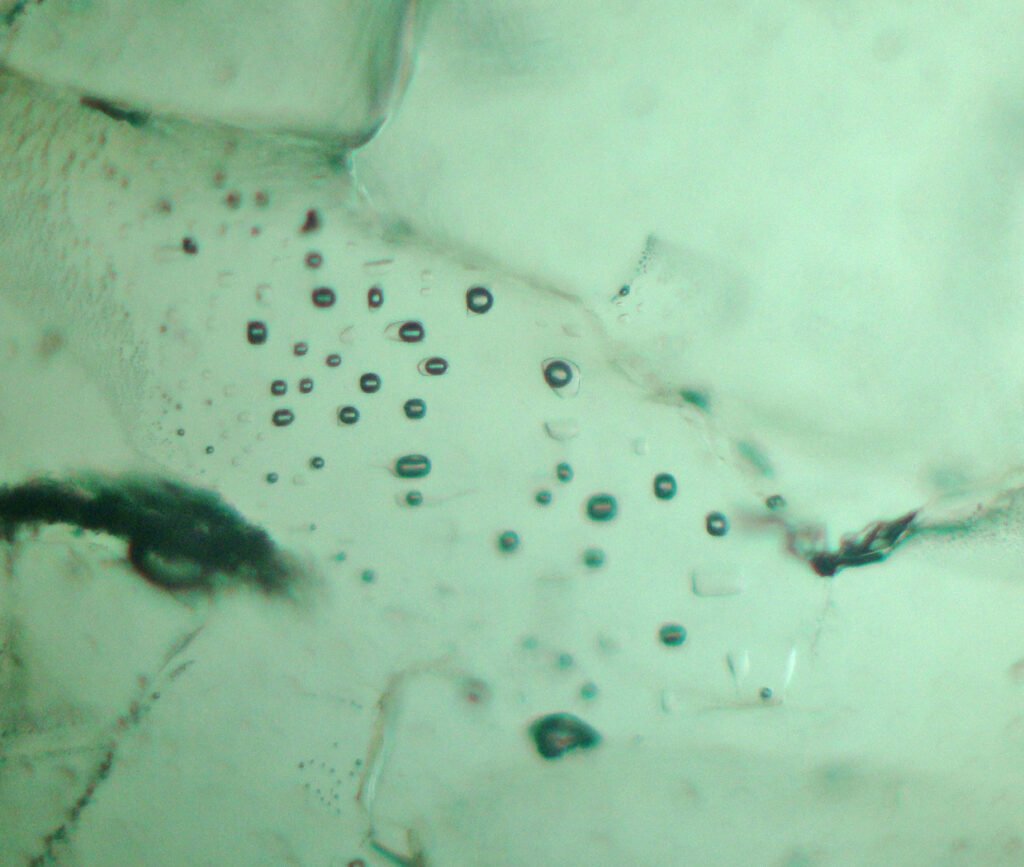
Halite, in the form of crystals, including cubic ones, occurs in polyphasic fluid inclusions of several different minerals. May be included in other medium metamorphic grade minerals.
It occurs as volcanic sublimates, as efflorescence and in caves.
3. Mineral Associations
In evaporites it occurs with gypsum, anhydrite, sylvite, carnallite, clay minerals, polyhalite, kieserite and kainite.
It also occurs with leonite, nahcolite, trona, picromerite and burkeite.
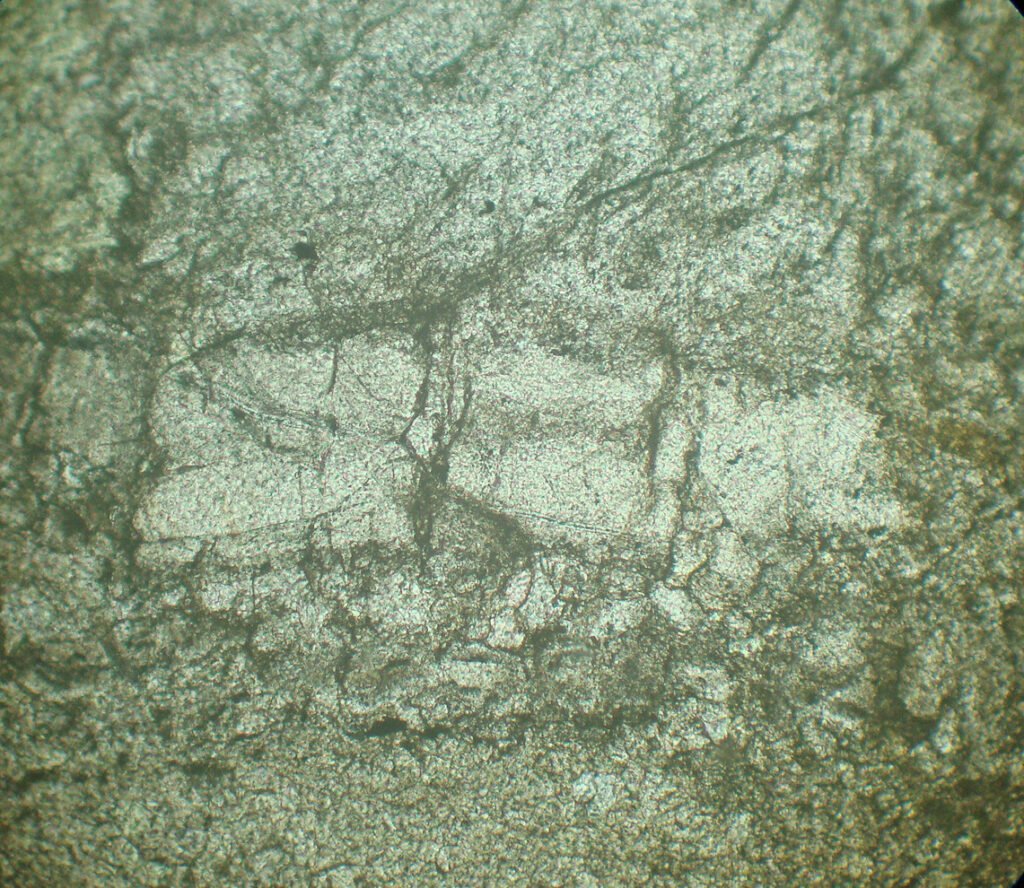
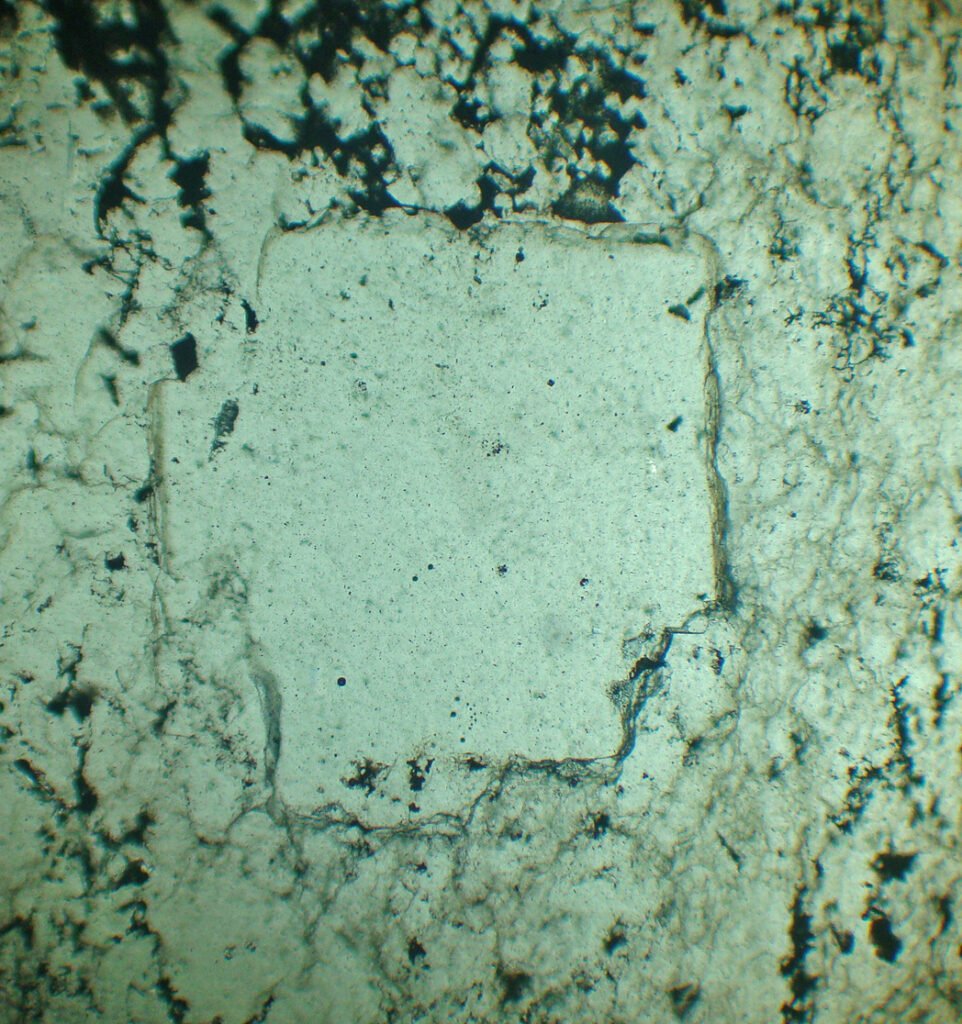
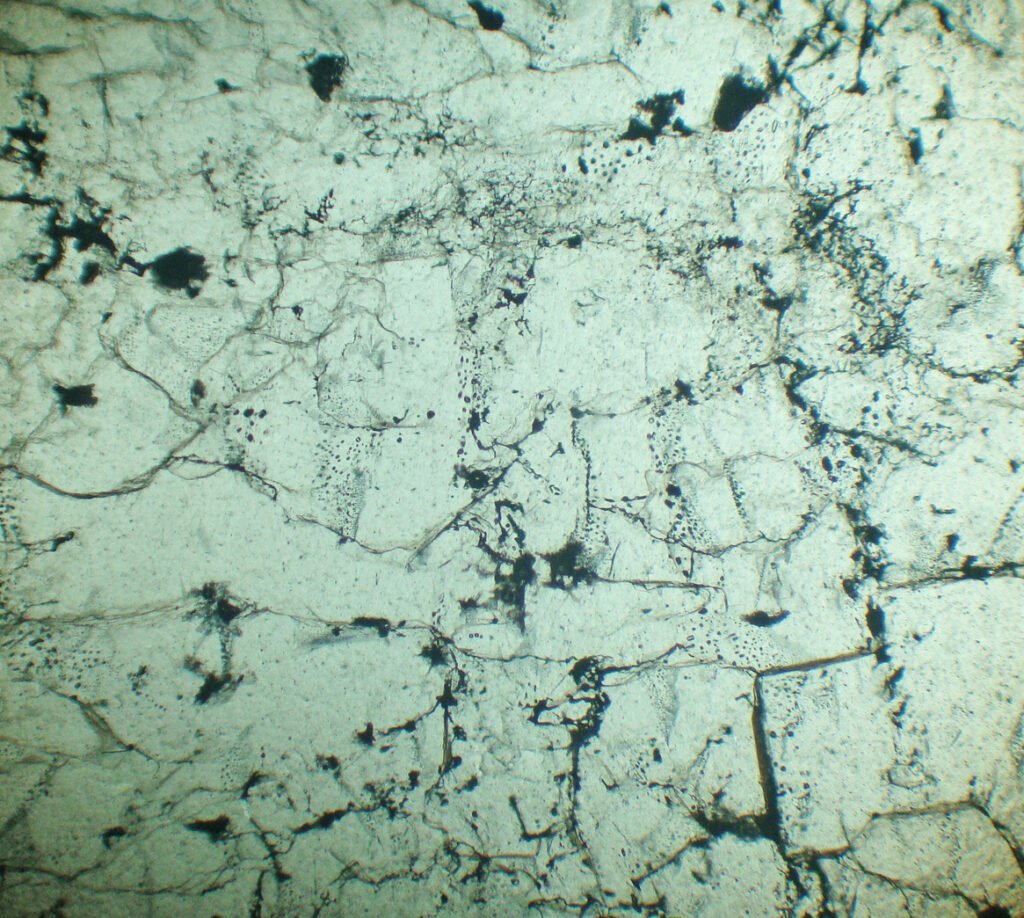
4. Transmitted Light Microscopy
The making of thin sections of evaporites, made up of minerals, many of which are soluble in water, needs to be carried out with special care, using mineral oil during grinding, which makes the process very difficult.
Once the slide is ready, it needs to be washed with alcohol and immediately covered with a coverslip to prevent the moisture in the air from dissolving the halite. Even so, it is possible that the moisture in the air dissolves the halite gradually, on a scale of months and years, penetrating laterally in the thin section, between the slide and the coverslip, along the layer of halite crystals.
Refraction indices: n: 1.5443
PLANE POLARIZED LIGHT – PPL
Color / Pleochroism: Colorless. If the salt has color, it may show weak pleochroism due to the stresses it has undergone.
Relief: Low.
Cleavage: Usually is not visible.
Habits: Cubes, massive.
CROSSED POLARIZED LIGHT – XPL
Birefringence and Interference Colors: Isotropic. May be weakly anisotropic due to stresses.
Extinction: Isotropic.
Elongation sign: Isotropic.
Twins: Isotropic.
Zoning: Isotropic.
CONVERGENT LIGHT
Character: Isotropic.
2V angle: Isotropic.
Alterations: No information available.
May be confused with: as halite is the only common mineral of evaporitic sequences that is cubic, its isotropy is highly diagnostic.


5. Reflected Light Microscopy
Does not applies.
