RHODOCHROSITE
Rhodochrosite – MnCO3 – is a rarer carbonate. It is a very secondary ore of Mn and has many uses as a decorative stone, fetching high prices in the collector and ornamental stone market, but it is not suitable for stoning. Once exposed to the atmosphere, it can develop a thin black surface film of Mn oxides. Manganese carbonate is extremely destructive to the amalgamation process used in the concentration of silver ores. For this reason, rhodochrosite was, for a long time, discarded in the tailings piles until it received acceptance as an ornamental stone. There are over 20 discredited names for rhodochrosite.
Pure rhodochrosite is very rare, the mineral always has Fe, Zn, Co, Mg, Cd and Ca contents. The red to pink color occurs in those crystals that have a minimal iron content; with increasing iron content the color changes to brown. It forms a continuous solid solution with calcite and another with siderite. Shows slow effervescence in cold HCl. Sometimes it fluoresces dark red. It has 5 varieties.
It can be confused with rhodonite, which is a manganese silicate with an almost identical color. Rhodochrosite and rhodonite even occur together, in fine granular aggregates, in sedimentary deposits.
1. Characteristics
Crystal system: Trigonal scalenohedral.
Color: Ranges from almost colorless to pink, red, yellow, cinnamon brown, may be banded. Black when altered.
Habit: Rhombohedral and scalenohedral crystals, up to 15 cm. Generally laminar, columnar, stalagtitic, botryoidal, granular, compact, massive.
Cleavage: {10-10} perfect.
Tenacity: Brittle.
Twinning: Common on {10-12}.
Fracture: Irregular, conchoidal.
Mohs Hardness: 3.5 – 4
Parting: On {10-12}, sometimes.
Streak: White.
Lustre: Vitreous, pearly on aggregates.
Diaphaneity: Transparent.
Density (g/cm³): 3.7
2. Geology and Deposits
Rhodochrosite is found as a primary mineral in low to moderate temperature hydrothermal veins containing native silver, native copper and lead sulfides, where it occurs with other Mn minerals. It occurs in high temperature metamorphic and metasomatic deposits and is common in carbonatites.
It is typical for sedimentary Mn deposits, where it accompanies Mn ore minerals. It can be autigenic and secondary in sediments. It rarely occurs as a late mineral in granitic pegmatites, especially in those with lithiophilite.
3. Mineral Associations
In hydrothermal occurrences it is associated with other carbonates (calcite, siderite, dolomite), other Mn minerals (rhodonite, tephroite), typical gangue minerals (fluorite, barite, quartz), sulfides (pyrite, chalcopyrite, galena, tetrahedrite, sphalerite ), native elements (gold, silver), tellurides and huebnerite.
In metamorphic occurrences it occurs with rhodonite, garnet and several other Mn minerals (alabandite, hausmannite, braunite, manganite, friedelite, etc.). In peralkaline rocks and associated pegmatites it occurs with aegirine and many dozen rare minerals (Mont Saint Hilaire, Quebec, Canada).
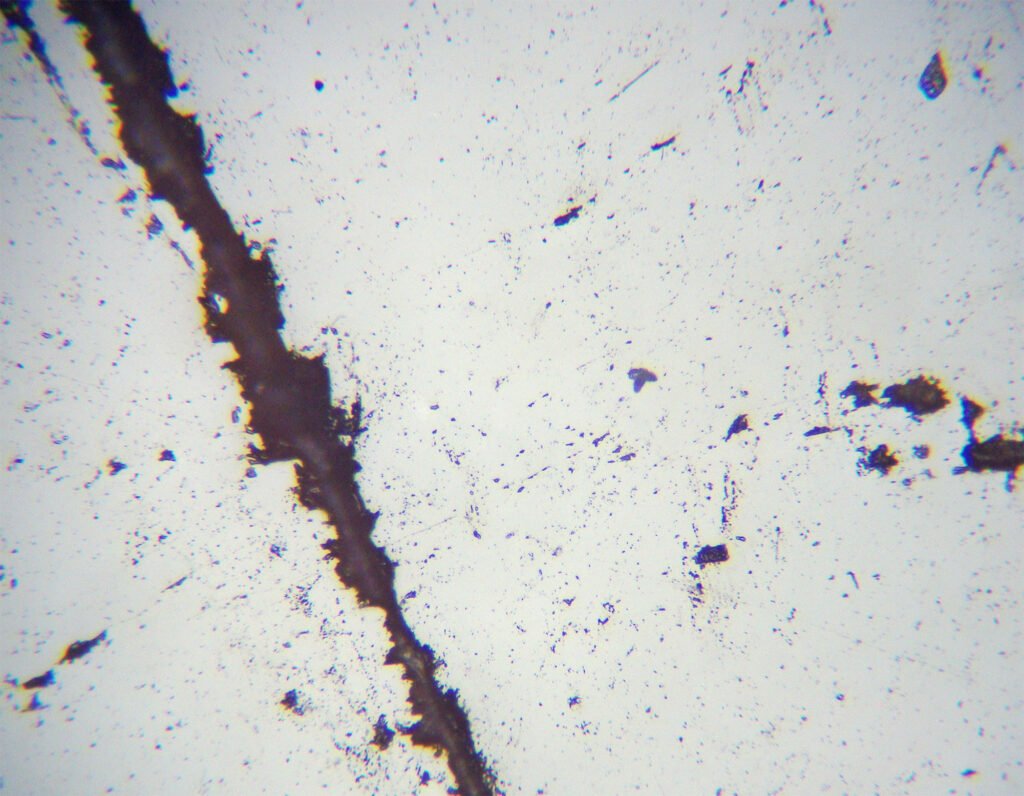
4. Transmitted Light Microscopy
Refraction indices: nω: 1,8175 nε: 1,597 (when pure!)
PLANE POLARIZED LIGHT – PPL
Color / Pleochroism: colorless to pale pink. May have weak pleochroism.
Relief: The relief varies between low and moderate to high at every 90º to the rotation of the stage in crystals with well defined cleavage. This phenomenon has been dubbed “relief pleochroism” or “chagrin change” and is typical of carbonates (calcite, dolomite, aragonite, siderite, rhodochrosite and magnesite).
When microcrystalline, these carbonates do not show “relief pleochroism”.
Cleavage: {10-11} perfect. This is the rhombohedral cleavage of carbonates, also present in calcite, dolomite, siderite and magnesite. The two cleavages intersect at angles of 60 and 120 degrees. There is a partition on {01-12}.
Habits: Lamellar aggregates. It can be columnar, stalagtite, botryoidal, compact, granular or massive.
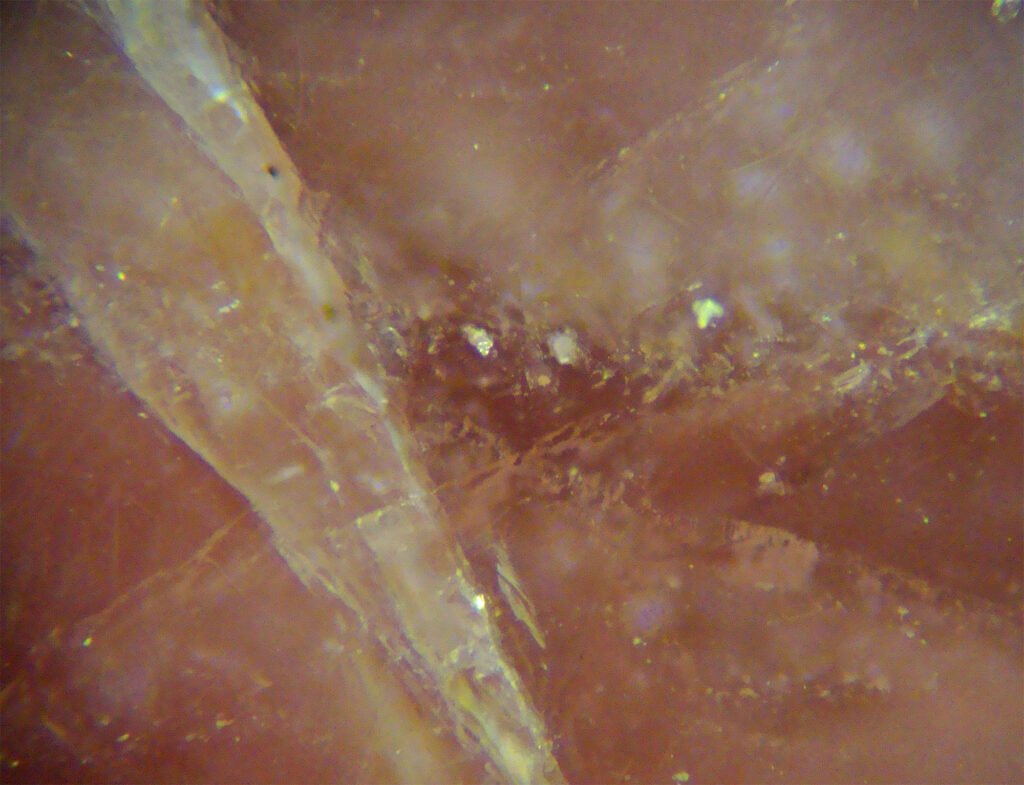
CROSSED POLARIZED LIGHT – XPL
Birefringence and Interference Colors: Maximum birefringence of 0.218, corresponding to very high interference colors, difficult to determine (“pearlaceous white”).
Extinction: Symmetrical with respect to the rhombohedral cleavage planes.
Elongation sign: Not applicable.
Twins: Contact and lamellar twins may occur.
Zoning: No.
CONVERGENT LIGHT
Character: U(-)
2V angle: No.
Alterations: through weathering and oxidation in supergene environments, rhdochrosite breaks down into various manganese oxide and hydroxide minerals like pyrolusite, manganite, psilomelane (Wad) and rhodocrosite-iron oxides. Hydrothermal alteration produces rhodonite and hausmannite.
May be confused with: the rhombohedral cleavage and high interference colors identify the mineral as a carbonate, but the carbonates cannot be distinguished from one another under a petrographic microscope; other analytical techniques are required.



5. Reflected Light Microscopy
Reflected light microscopy is not the recommended analytical method for the identification of rhodochrosite. However, it is important to make a polished thin section or a polished section to identify the opaque minerals that occur associated with rhodocrosite.
Sample preparation: rhodochrosite acquires a very good polish. Its polishing hardness is superior to that of siderite, but inferior to that of smithsonite. The properties vary greatly depending on the Fe, Mg and Ca contents.
PLANE POLARIZED LIGHT – PPL
Reflection color: Dark gray.
Pleochroism: No.
Reflectivity: Very low (<<10%)
Bireflectance: Very weak.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: It does not show anisotropy.
Internal reflections: White, strong multicolored and strong pink internal reflections. These internal reflections do not always allow to recognize the pink color of the mineral.
May be confused with: difficult to recognize, but indicative are the association with Mn minerals, the color and the slow reaction with dilute hydrochloric acid. Siderite and smithsonite are very similar, but the black alteration products (of Mn) aid in the identification of rhodochrosite.
General Characteristics:
Triangular polishing pits may be present due to the excellent cleavage of rhodochrosite. These figures are very rare due to the excellent polish that rhodochrosite acquires. In addition, the figures are much less defined than those of galena, for example. They are irregular alignments of more or less geometric holes, often difficult to define.
Substitutions, extensive and conspicuous, by pyrolusite, “psilomelane” and other Mn oxides can occur and are very diagnostic.
Textures are similar to those of siderite.