SILVER
Silver – Ag – is a rarer native element, very important as a silver ore. The metal has a multitude of uses and a huge and growing number of industrial applications. Dendritic silver pieces are worth much more in the collector minerals market than their weight in metal.
Silver is classified in the Copper Group. Can form crystals mixed with gold in any proportion (electrum), but alloys with percentages of 0-1% Au, ~20% Au and ~60% Au are especially frequent. May contain Hg (up to 20%), Cu (up to 1%), Bi (up to 8%), Sb (up to 5%) (Dyscrasite), As, Pt, Ni, Pb and Fe. It forms a series with Au and another series with Pd. There are a dozen varieties based on the content of other elements.
Crystals are very rare and form cubes, octahedrons and dodecahedrons of no more than 2 cm; can form parallel clusters. Silver always tarnishes to black due to the formation of an acanthite film.
1. Characteristics
Crystal system: Cubic hexaoctahedral.
Color: Silvery white. Forms a pink to cream film, tarnishes to gray and black (acanthite).
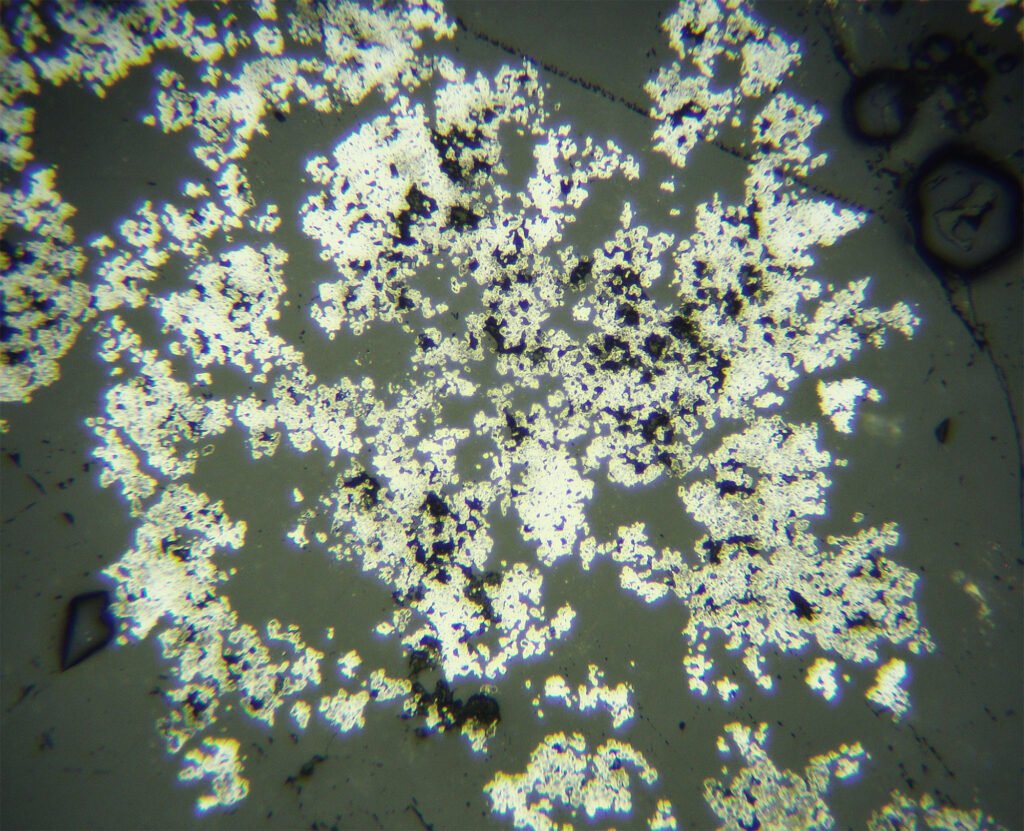
Habit: Filiform, capillary, plates, dendritic, massive, “herringbone”. Crystals very rare.
Cleavage: No.
Tenacity: Ductile, malleable.
Twinning: On {111} and {100}, interpenetration.
Fracture: No.
Mohs Hardness: 2.5 – 3
Parting: No.
Streak: Silvery white.
Lustre: Metallic, intense.
Diaphaneity: Opaque.
Density (g/cm³): 10.1 – 11.1
2. Geology and Deposits
Silver occurs as a primary mineral in volcanic rocks (andesites) and mainly in sulphide mesothermal hydrothermal veins, in the Ni-Co-As-Ag-Bi association, usually with U (Caution required!).
As a secondary mineral, it forms in the oxidation zones of sulphide ores.
Finally, through weathering and erosion, it can accumulate in placers in the form of nuggets.
The macro- and microscopic habits of silver, both hydrothermal and secondary, can be extremely similar, requiring great care in the interpretation of the generated textures.
3. Mineral Associations
In hydrothermal veins, silver occur associated with quartz, carbonates (calcite, siderite), native elements (copper, arsenic), other Ag minerals (acanthite, pyrargyrite, proustite, dyscrasite), chalcocite, arsenopyrite, allargentum, safflorite, cobaltite, uranium minerals and zeolites.
In the oxidation zones (“iron hat”), it occurs with goethite, anhydrite, chalcocite, galena, secondary silver minerals (acanthite), secondary Cu minerals (cuprite, etc.) and sphalerite, among others.
In sedimentary deposits, it occurs in shales (“Kupferschiefer”), “red beds” and sandstones.
It is also associated with Co and Ni arsenides, chlorargyrite, embolite, silver sulphosalts, native bismuth, breithauptite, calaverite, dyscrasite, graphite, stromeyerite, freibergite, tetrahedrite-tennantite and others.
4. Transmitted Light Microscopy
Not applicable, as silver is completely opaque.
5. Reflected Light Microscopy
Sample preparation: polishing pure silver is easy, but care is needed and only very fine abrasives should be used so as not to generate deep polishing scratches. The formation of holes is also common during polishing. The polishing hardness is:
– greater than the hardness of galena and proustite,
– slightly greater, almost equal, to the hardness of chalcopyrite. A little less than that of dyscrasite.
– lower than the hardness of arsenic and tetrahedrite-tennantite. Much lower than that of sphalerite.
PLANE POLARIZED LIGHT – PPL
Reflection color: Light silver-white, glossy, sometimes a little cream.
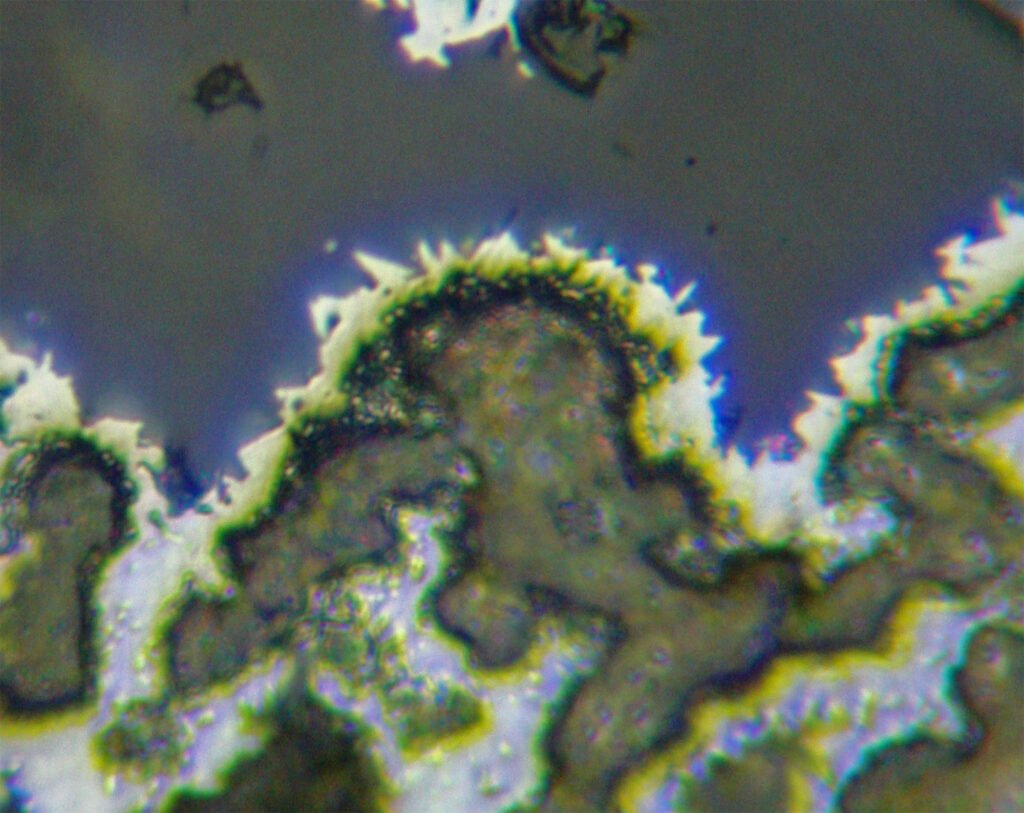
Air corrosion is weak to distinct; shades of yellow, also red, blue and other colors appear due to the formation of a superficial film of acanthite (Ag2S).
This tarnishing occurs at very different speeds in grains of different occurrences; sometimes it is very fast and appears during the observation of the section with reflected light.
Polished sections with strong tarnish look like tarnished copper.
Compared with the color of copper, the color of silver is white and lighter.
Compared with the color of platinum, the color of silver is cream white and lighter.
Compared with the color of bismuth, the color of silver is almost the same, somewhat lighter.
Compared with the color of arsenic, the color of silver is much lighter and creamier.
Compared with the color of antimony, the color of silver is lighter and cream-white.
Pleochroism: No.
Reflectivity: 82.82%. If with Sb, 87.5%. Extremely high! It is the highest in Reflected Light, dazzles the observer, is an important diagnostic aspect!
Bireflectance: No.
CROSSED POLARIZED LIGHT – XPL
Isotropy / Anisotropy: Isotropic, but the many polishing scratches give the mineral the impression of anisotropy and generate other wrong optical effects.
Internal reflections: No.
May be confused with: in freshly polished sections silver can only be confused with minerals of very high reflective power. On old and tarnished sections great care must be taken.
Native bismuth is softer and anisotropic.
Native antimony does not tarnish (but sometimes silver does not), can show cleavage, and is slightly anisotropic.
Dyscrasite is easy to confuse with silver.
Native platinum is very similar, but is less yellow, with the exception of antique polished sections.
Pyrite is much harder and has much lower reflectivity.
General Characteristics:
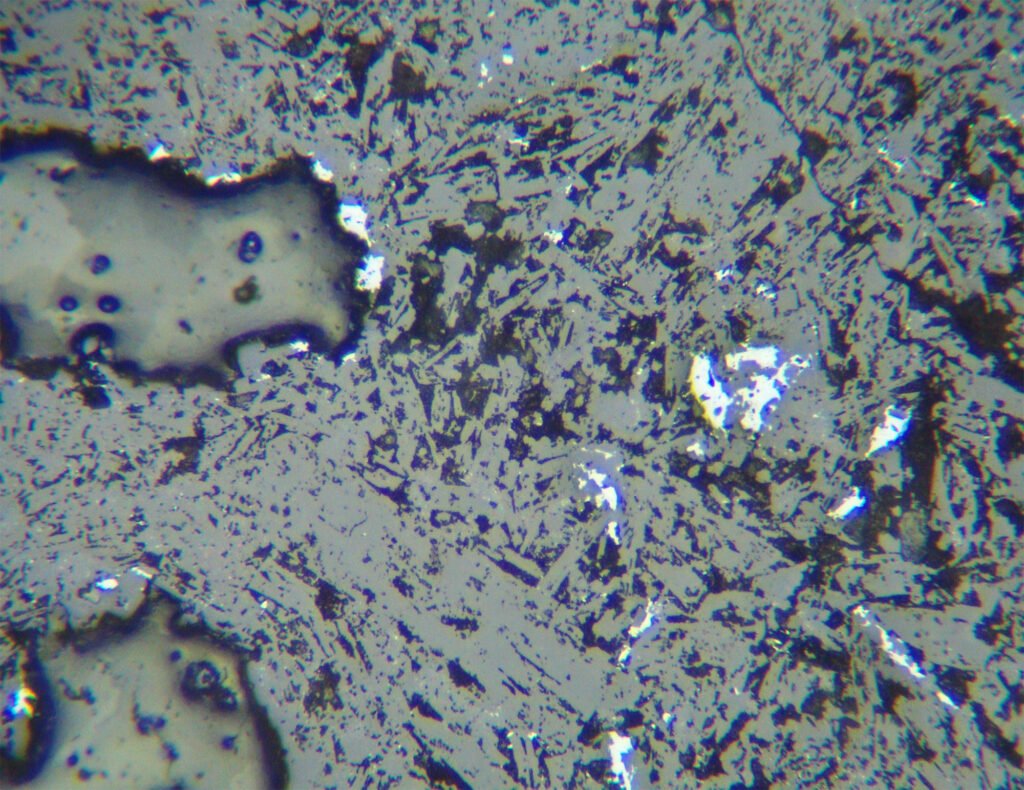
Grain shape varies greatly. Euhedral grains are quite rare, usually silver form dendrites and skeletons. “Knitted silver” is silver enveloped by nickeline, rammelsbergite or skutterudite-Ni. Silver can form irregular masses, scattered grains, crusts over other minerals, etc. Crack fillings (cleavages, discontinuities in general, intergranular boundaries and cataclastic zones) and thin surface films are observed in silver deposited in the cementation zone.
Polishing scratches, usually fine, are often present and difficult to eliminate. It is not worth insisting on the perfect polishing of silver because there is a risk of producing a very uncomfortable negative relief of silver in relation to the other minerals present, due to the lower hardness of silver.
Twins are relatively common, lamellar, can be of growth or pressure, especially when the silver is present in coarser grains. The lamellae are often deformed.
Zonation occurs sometimes, may be well developed. In some instances, the silver grains have porous surface films, which may be due to previous acanthite encrustation.
Unmixing (exsolution) features are usually absent. Some occur with dyscrasite and with allargentum.
Decomposition of acanthite, freibergite or galena-Ag can form silver.
Silver intergrowths occur with bornite, chalcocite and galena.
Substitutions 1: silver can replace sphalerite, chalcopyrite, galena, silver minerals (acanthite), Cu minerals (bornite, chalcocite), nickel, allargentum, bismuth, stephanite, pearceite, maucherite, stromeyerite, tetrahedrite-tennantite, arsenopyrite, pitchblende, native bismuth and zones in skutterudite.
Substitutions 2: silver is replaced by galena, acanthite, rammelsbergite, younger sulfides and arsenides.
Mirmequites, very well developed, can be generated by substitutions.
Pseudomorphoses of silver occur on pitchblende, acanthite, proustite, pyrargirite, stephanite, safflorite and rammelsbergite.
Inclusions of silver occur in acanthite, pyrargyrite, rammelsbergite, bornite, chalcopyrite, arsenic, dyscrasite and pyrrhotite.
Space fillings such as fractures, cracks, cataclastic zones and intergranular boundaries are frequent in secondary silver. They form associated with galena, sphalerite, tennantite-tetredrite, bornite and covellite.
The acanthite-silver pair has an intimate relationship: one transforms into the other and vice versa.
Disintegration of stromeyerite and of Ag-Sb mixture crystals forms textures that closely resemble the textures generated by the substitutions.
Eutectic intergrowths with native copper are extremely rare but do occur.